Pharmaceutical Management
Navigating complex regulations and ensuring quality in pharmaceutical manufacturing? ChecklistGuro's Work OS platform streamlines processes from R&D to production, packaging, and distribution. Maintain compliance, improve efficiency, and reduce risk. Discover how ChecklistGuro can transform your pharmaceutical operations!
No credit card or cancellation needed.
Improved Compliance Adherence
Increased Operational Efficiency
Enhanced Data Integrity
Centralized Control for Unparalleled Compliance
Pharmaceutical regulations are complex and constantly evolving. Our solution centralizes all your compliance documentation, SOPs, training records, and audit trails in a single, accessible Work OS. Reduce risk, streamline audits, and ensure adherence to cGMP, FDA, and other critical standards. Automated workflows and real-time alerts keep everyone on the same page.
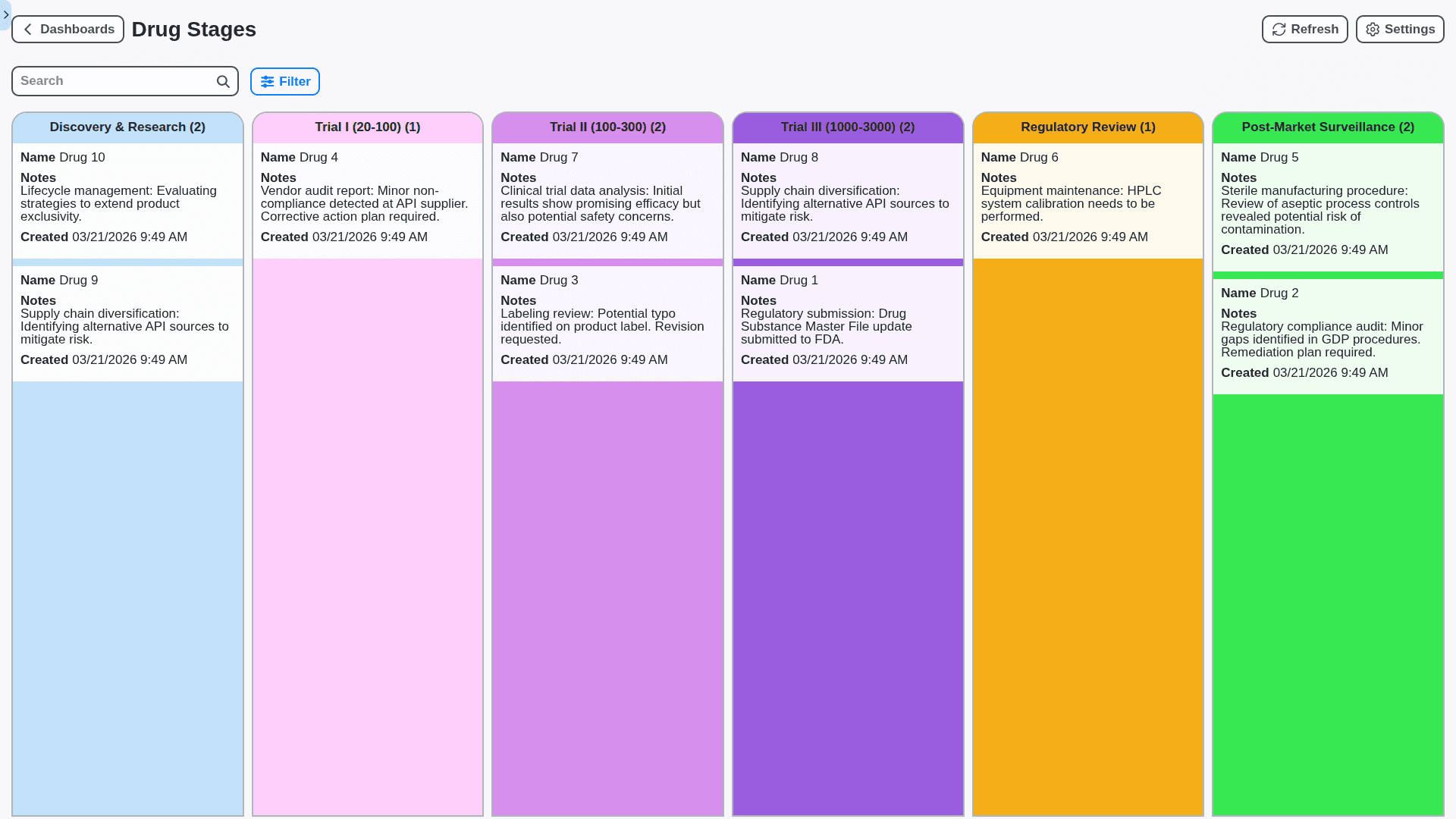
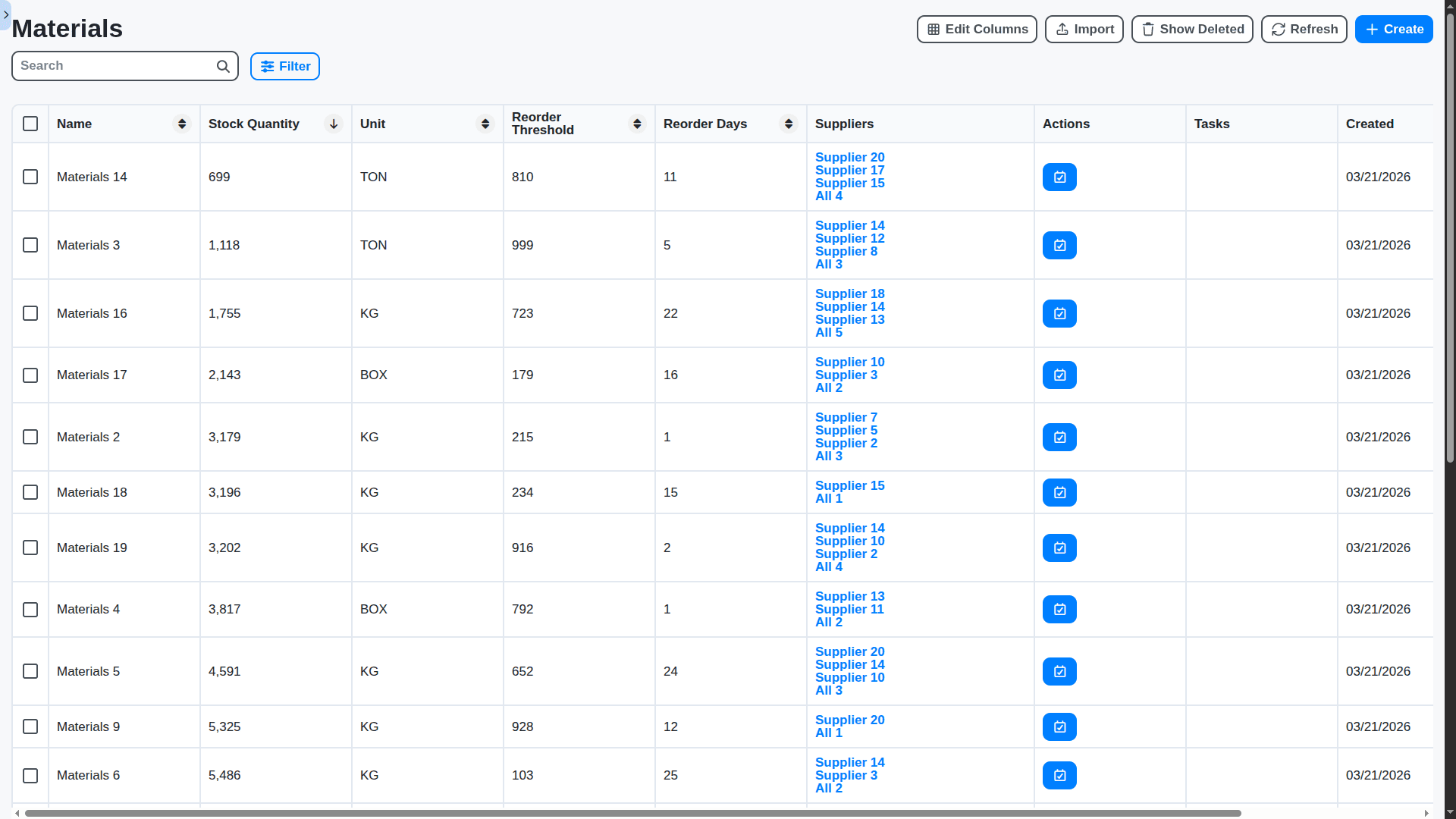
Real-Time Inventory Visibility & Optimized Stock Levels
Eliminate stockouts and minimize waste with a real-time view of your pharmaceutical inventory across all locations. Track lot numbers, expiry dates, and temperature conditions. Our Work OS provides automated reorder points, demand forecasting, and detailed reporting to optimize inventory levels and reduce costs.
Seamless Collaboration Across Teams & Departments
Break down silos and improve communication. Our Work OS connects pharmacy, quality, manufacturing, and distribution teams, fostering collaboration and ensuring everyone has access to the information they need, when they need it. Automated notifications, task assignments, and shared workspaces streamline workflows and reduce errors.
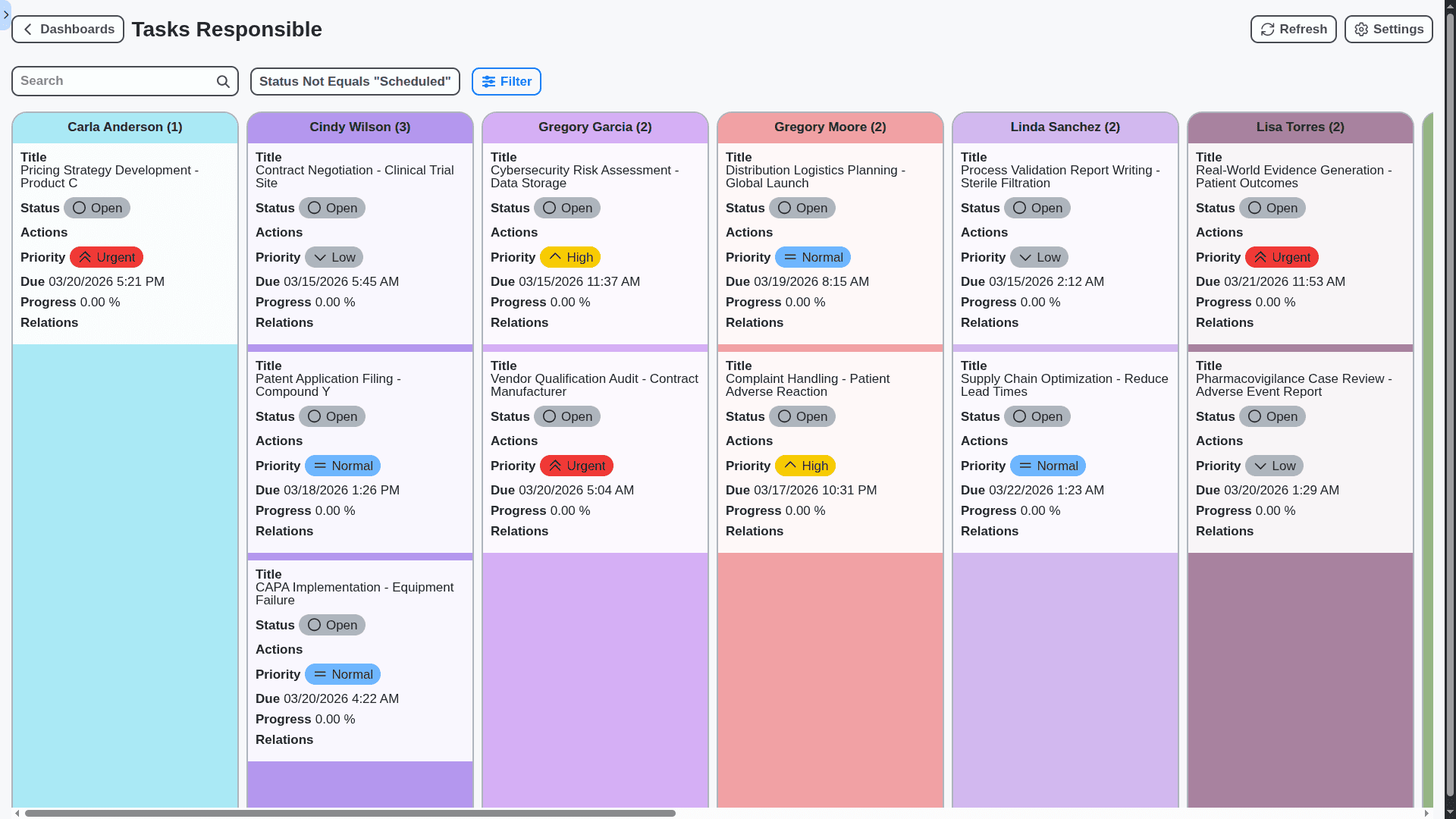
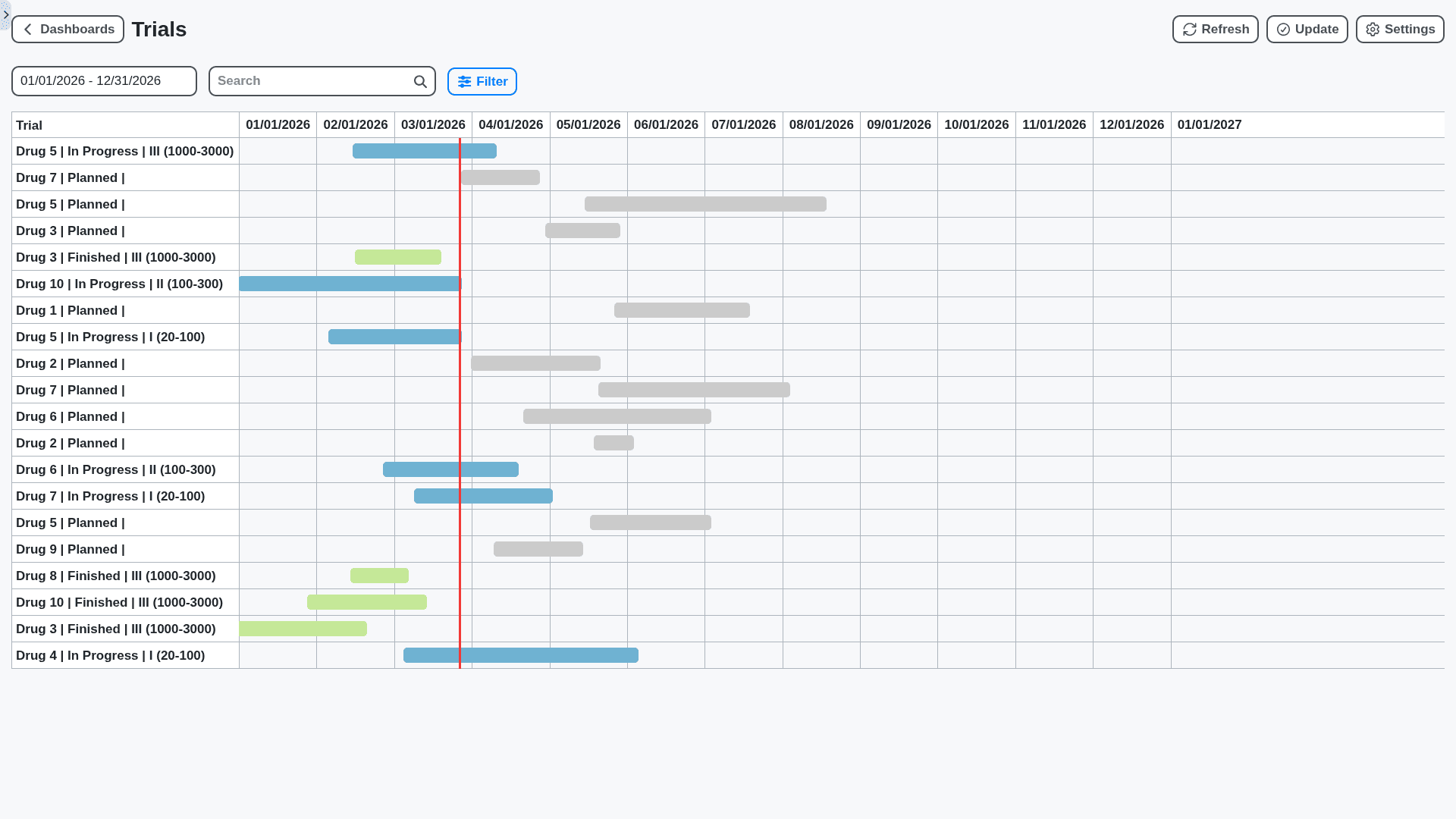
Your Business, Your Workflow: Fully Customizable Pharmaceutical Management
Forget rigid systems! Our Work OS offers unparalleled customization. Tailor dashboards, workflows, forms, and reports to perfectly match your unique processes, team structures, and regulatory requirements. Configure everything from user roles to data fields, ensuring a truly bespoke solution.
Adaptable Workflows: From Pharmacy to Manufacturing & Beyond
Whether you're managing a small pharmacy, a large-scale manufacturing plant, or a complex distribution network, our Work OS adapts to your environment. Configure workflows to reflect your unique processes, regardless of size or complexity.
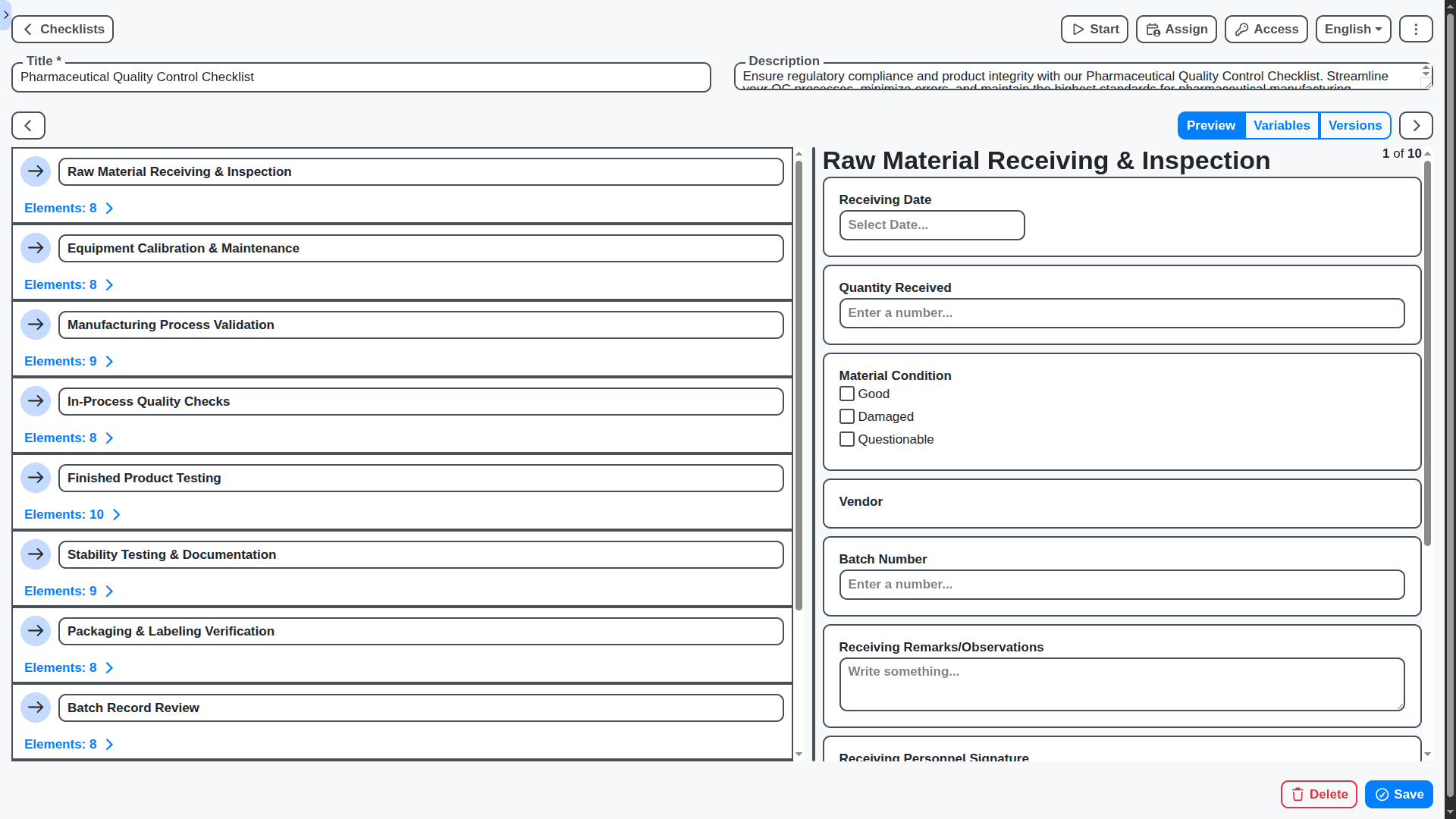
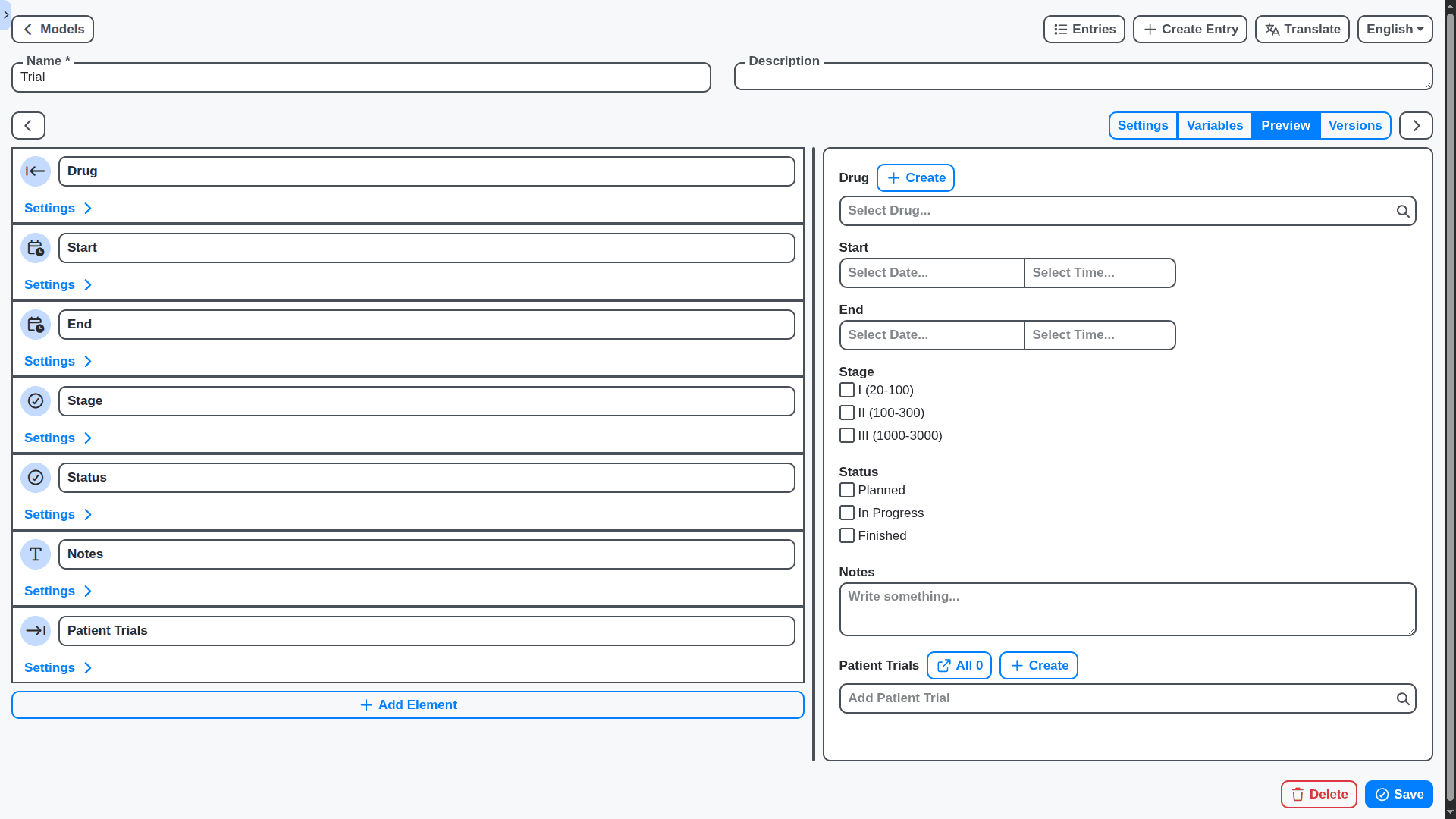
Data Fields That Work For You: Define Your Information
Standard data fields often don't capture everything you need. Our Work OS allows you to create custom data fields to track exactly the information that's important to your business. Add new fields to forms, reports, and dashboards with ease, ensuring you have a complete picture of your operations.
Role-Based Permissions: Control Access with Precision
Ensure data security and compliance by defining granular role-based permissions. Tailor user access to specific modules, data fields, and workflows, ensuring that only authorized personnel can access sensitive information.

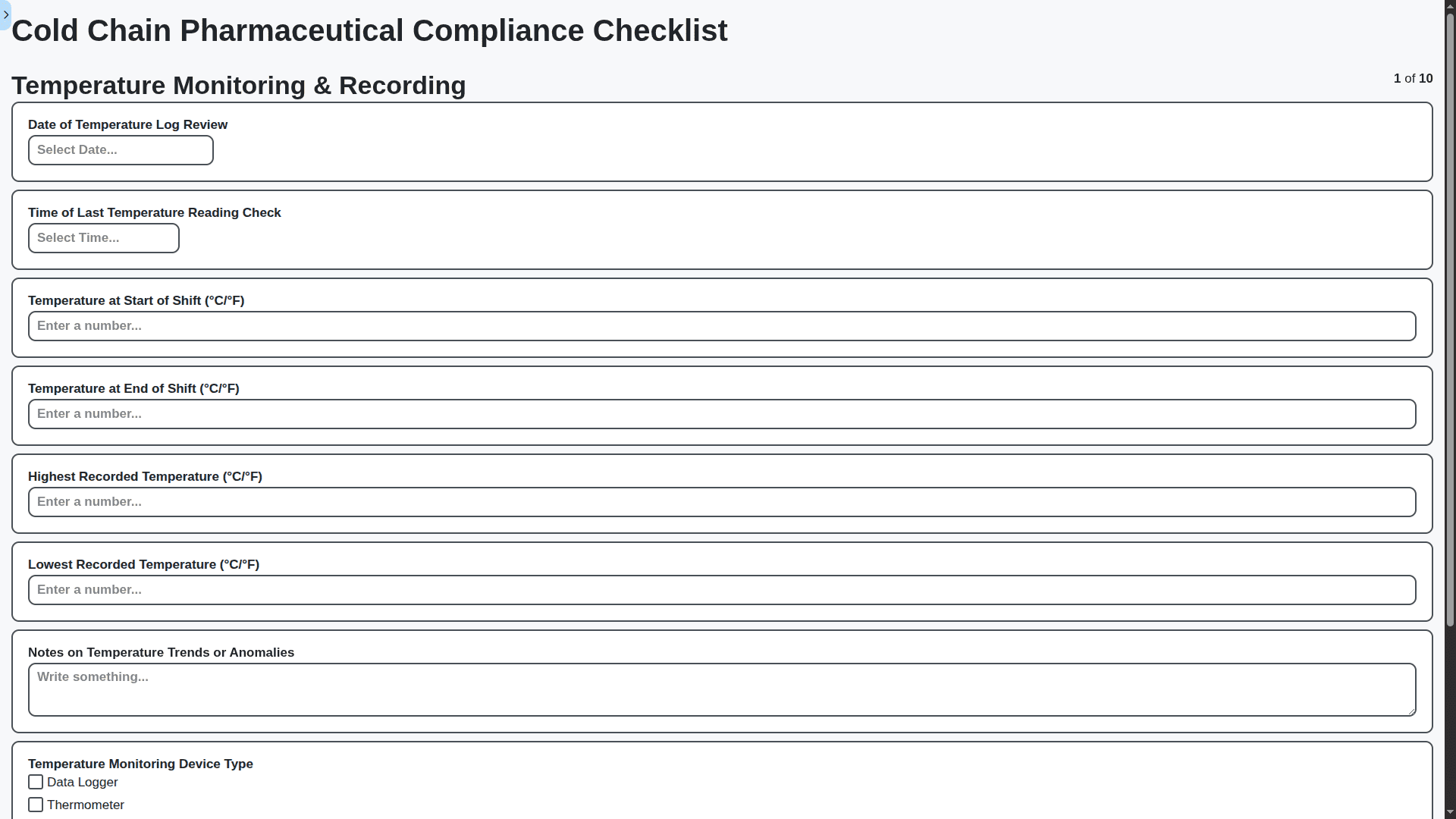
Digitize Standard Operating Procedures (SOPs) & Training
Replace cumbersome paper-based SOPs with a dynamic, digital solution. Our platform centralizes SOPs, automates approvals, enforces version control, and simplifies training and competency management. Reduce errors, improve compliance, and empower your workforce with easy access to critical information.
No credit card or cancellation needed.
Features of our Pharmaceutical Management Solution
Core Work OS Capabilities
Dynamic Workflow Modeling: Visual, drag-and-drop interface for designing complex, adaptable workflows.
Process Simulation & Optimization: Predictive analysis and simulation to identify bottlenecks and improve process efficiency. Includes "what-if" scenario planning.
Real-time Process Monitoring & Dashboards: Interactive dashboards displaying key process metrics, KPIs, and alerts. Customizable views for different roles.
Automated Task Assignment & Escalation: Intelligent routing of tasks based on pre-defined rules and conditions. Automated escalation paths for overdue tasks.
Business Rules Engine: Centralized rules repository for managing and enforcing regulatory compliance and business logic. Rules are versioned and auditable.
Mobile Access: Access and manage workflows and tasks from anywhere, on any device (iOS, Android). Offline capabilities included.
Integration Platform: Pre-built connectors and API support for seamless integration with existing systems (ERP, CRM, LIMS, MES, etc.).
Reporting & Analytics: Comprehensive reporting capabilities with customizable templates and data visualizations. Trend analysis and predictive reporting.
Form Builder: No-code form builder for creating dynamic, data-driven forms to capture information within workflows.
Collaboration Tools: Integrated communication features (chat, comments, document sharing) for enhanced collaboration between process participants.
Clinical Trial Management
eTMF (Electronic Trial Master File) Integration: Secure storage, versioning, and audit trail for clinical trial documents.
Investigator Site Management: Centralized portal for managing investigator sites, contracts, and payments.
Patient Recruitment & Enrollment: Workflow automation for patient identification, screening, and enrollment. Privacy-compliant data handling.
Adverse Event Reporting: Automated workflows for collecting, validating, and reporting adverse events. Integration with regulatory reporting systems.
Clinical Data Management (CDM) Integration: Connectors to leading CDM systems.
Protocol Deviation Management: Workflow for identifying, documenting, and resolving protocol deviations.
Drug Manufacturing & Quality Control
Batch Record Management: Electronic batch records with full audit trail and versioning.
Deviation Management: Workflow for investigating, documenting, and resolving manufacturing deviations. Root cause analysis tools included.
Change Control Management: Automated change request workflow with impact assessment and approval processes.
Corrective and Preventive Actions (CAPA): Workflow for tracking and resolving CAPA items.
Equipment Management & Maintenance: Integration with CMMS (Computerized Maintenance Management System) for equipment maintenance and calibration tracking.
Quality Risk Management (QRM): Workflow support for FMEA (Failure Mode and Effects Analysis) and other QRM activities.
Validation Management: Workflow automation for equipment and process validation activities.
Release Management: Automated workflows for drug product release.
Supply Chain Management
Demand Forecasting & Planning: Integration with S&OP (Sales and Operations Planning) systems.
Inventory Management: Real-time visibility into inventory levels across the supply chain.
Order Management: Automated order processing and tracking.
Serialization & Track & Trace: Compliance with serialization regulations (e.g., DSCSA).
Supplier Management: Workflow for managing supplier contracts and performance.
Regulatory Compliance & Reporting
21 CFR Part 11 Compliance: Secure electronic records and electronic signatures with full audit trails.
GxP Compliance: Workflow automation to support GxP regulations (GMP, GCP, GLP).
eCTD Submission Management: Automated workflows for creating and submitting electronic Common Technical Document (eCTD) submissions to regulatory agencies.
Periodic Safety Update Reports (PSUR) Management: Automated workflows to gather and analyze data for PSUR submissions.
Pharmacovigilance (PV) Integration: Seamless data exchange with PV systems.
No credit card or cancellation needed.
Frequently Asked Questions
What is ChecklistGuro and how can it help pharmaceutical companies?
ChecklistGuro is a Work OS platform designed to streamline critical pharmaceutical processes, reduce risk, and improve compliance. We help you automate SOPs, manage batch records, simplify validation, and enhance overall operational efficiency.
What types of pharmaceutical processes can ChecklistGuro manage?
ChecklistGuro can be used for a wide range of processes, including batch record management, SOP adherence, equipment validation, change control, quality control, deviation management, and more.
What are the key benefits of using ChecklistGuro for pharmaceutical management?
Key benefits include improved compliance adherence, increased operational efficiency, enhanced data integrity, reduced risk, and faster time to market.
How does ChecklistGuro help companies comply with regulatory requirements like FDA and EMA?
ChecklistGuro provides a centralized, auditable platform to manage processes and documentation, ensuring compliance with regulations like FDA, EMA, and ICH. Our platform supports built-in controls and comprehensive reporting to demonstrate adherence.
Is ChecklistGuro suitable for companies of all sizes within the pharmaceutical industry?
Yes! ChecklistGuro is flexible and scalable, making it suitable for pharmaceutical companies of all sizes, from small biotech startups to large multinational corporations.
How long does it take to implement ChecklistGuro?
Implementation time varies depending on the complexity of your processes and the number of integrations required. Most companies can see initial benefits within 4-8 weeks.
What level of IT expertise is required for implementation and ongoing management?
ChecklistGuro is designed to be user-friendly. While some IT involvement may be required for integration, most users can manage the platform with minimal training.
Can ChecklistGuro be customized to fit our specific pharmaceutical processes?
Absolutely! ChecklistGuro offers extensive customization options, allowing you to tailor the platform to your unique workflows and requirements.
Can ChecklistGuro be integrated with our existing systems (e.g., LIMS, ERP)?
Yes! ChecklistGuro offers a robust integration framework, allowing seamless data flow between our platform and your existing LIMS, ERP, and other critical systems.
What training and support are provided?
We offer comprehensive training programs, including online tutorials, live webinars, and on-site training options. We also provide ongoing technical support via email, phone, and online chat.
What are the system requirements for ChecklistGuro?
ChecklistGuro is a web-based application and can be accessed from any device with a modern web browser.
Is ChecklistGuro a cloud-based or on-premise solution?
ChecklistGuro is primarily offered as a cloud-based (SaaS) solution for ease of access and maintenance. There is also an option to install it on-premise, please contact us for more details.
How much does it cost?
We have 3 plans: Free Forever, Pro and Enterprise. Please check our pricing page for more details.
We can do it Together
Make a Request
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.