Food Manufacturing Inspection Workflow: HACCP & GMP Compliance Tracking
Streamline food safety inspections with our HACCP & GMP Compliance Tracking Workflow. Minimize risk, automate documentation, and ensure airtight food manufacturing standards on your Work OS Platform.
Start
Start of the Workflow/Process.
1. Retrieve Relevant Food Safety Standards
Automatically pull current regional and federal food safety regulations for checklist context.
2. Assign Inspection Task to Team Member
Assign the inspection task to the designated team member(s) based on roles.
3. Retrieve Product Batch Records
Fetch existing batch data to check for necessary documentation references.
4. Generate Digital Inspection Checklist
Create the actionable checklist tailored to the specific food category being inspected.
5. Record Temperature Logs (Critical Control Points)
Update specific data entries with measured temps (e.g., chiller units, cooking equipment).
6. Document Non-Conformance Issues
Create a new record when a potential health or safety violation is found during the walk-through.
7. Calculate Audit Score/Pass Rate
Execute a formula to determine overall compliance score based on filled-out checklist items.
8. Alert Manager on Failed Inspection
Send immediate email notification to the supervisor if critical fails are logged.
9. Send On-Site Reminder to Inspector
Send an SMS reminder to the inspector to complete an outstanding step or gather necessary signatures.
10. Generate Final HACCP Audit Report
Compile all data, photos, and findings into a comprehensive, time-stamped, and immutable report.
11. Verify Personnel Training Status
Check the system for required certifications for inspectors performing the task.
12. Approve/Reject Inspection Findings
Update the master record status after review, confirming remediation actions.
End
Start of the Workflow/Process.
Start of the Workflow/Process.
Automatically pull current regional and federal food safety regulations for checklist context.
Assign the inspection task to the designated team member(s) based on roles.
Fetch existing batch data to check for necessary documentation references.
Create the actionable checklist tailored to the specific food category being inspected.
Update specific data entries with measured temps (e.g., chiller units, cooking equipment).
Create a new record when a potential health or safety violation is found during the walk-through.
Execute a formula to determine overall compliance score based on filled-out checklist items.
Send immediate email notification to the supervisor if critical fails are logged.
Send an SMS reminder to the inspector to complete an outstanding step or gather necessary signatures.
Compile all data, photos, and findings into a comprehensive, time-stamped, and immutable report.
Check the system for required certifications for inspectors performing the task.
Update the master record status after review, confirming remediation actions.
Start of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
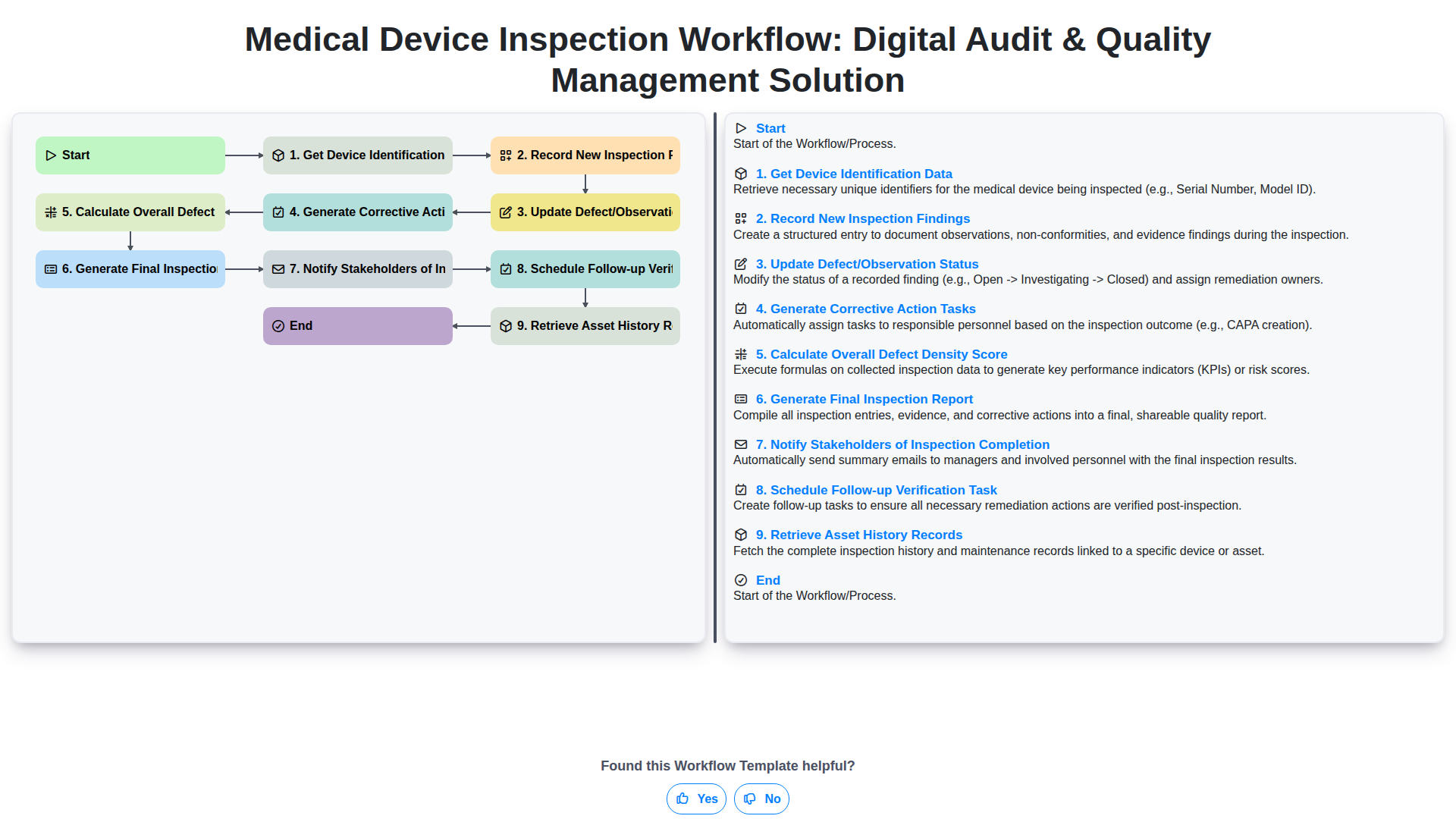
Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
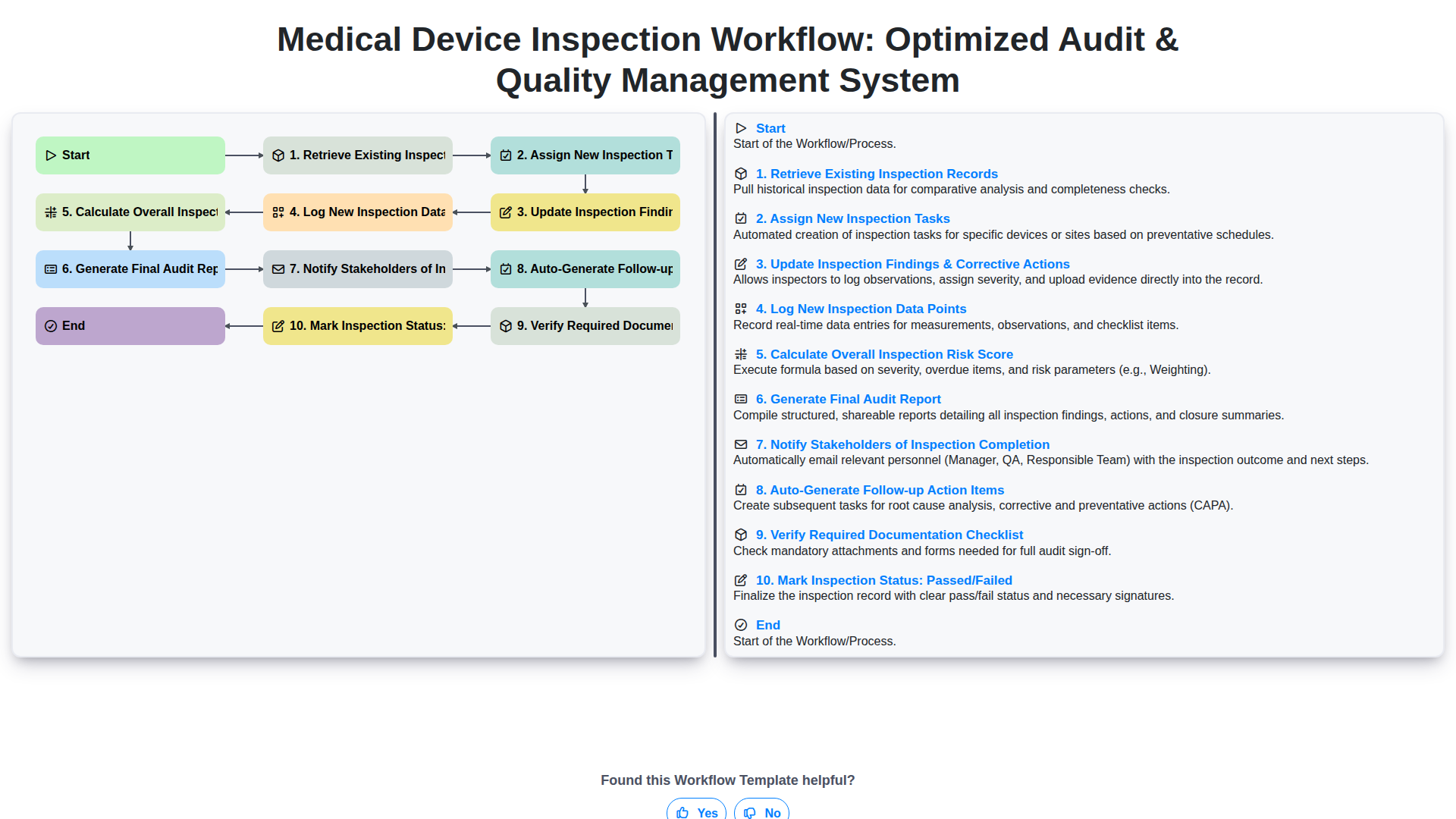
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
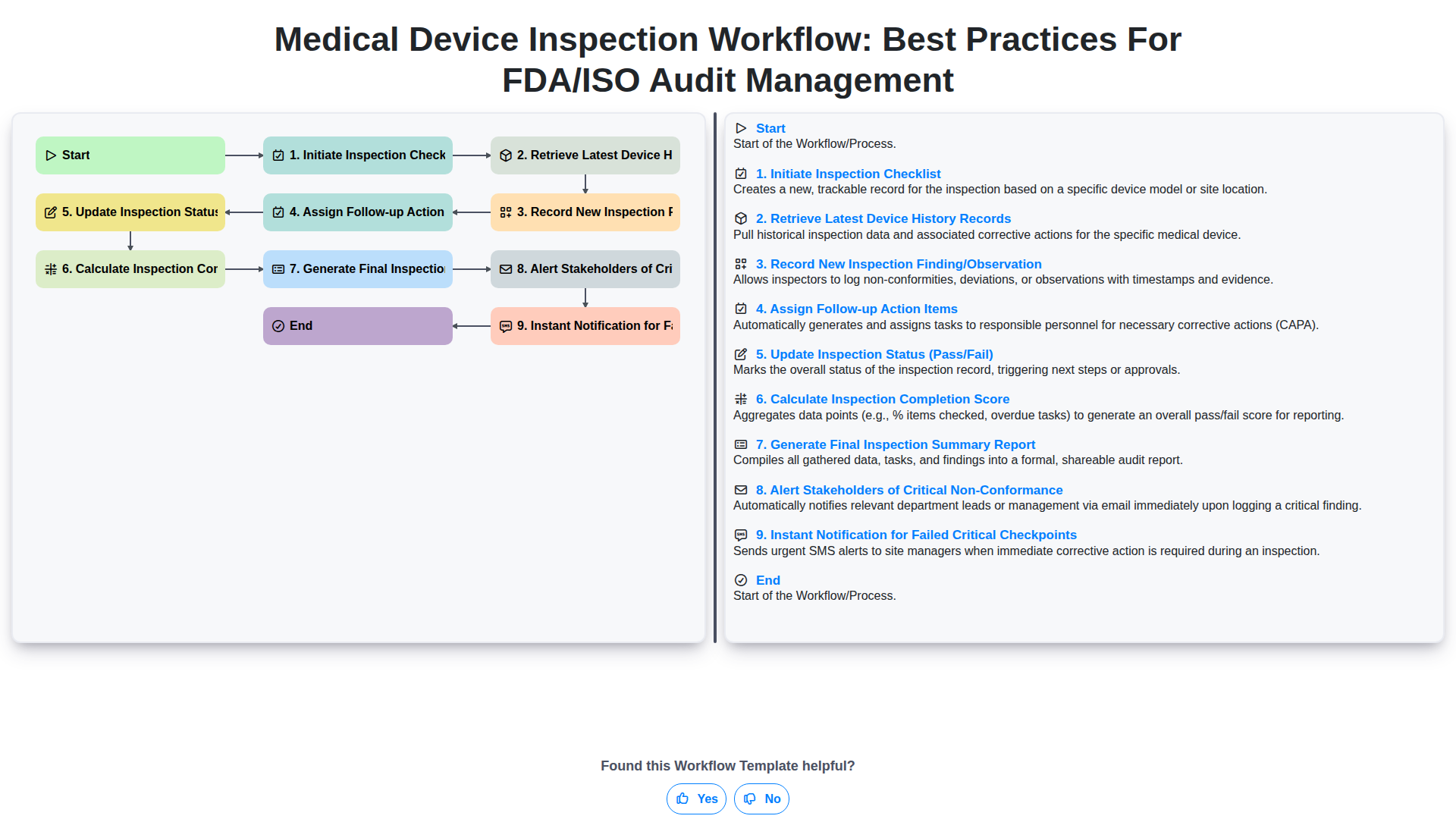
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
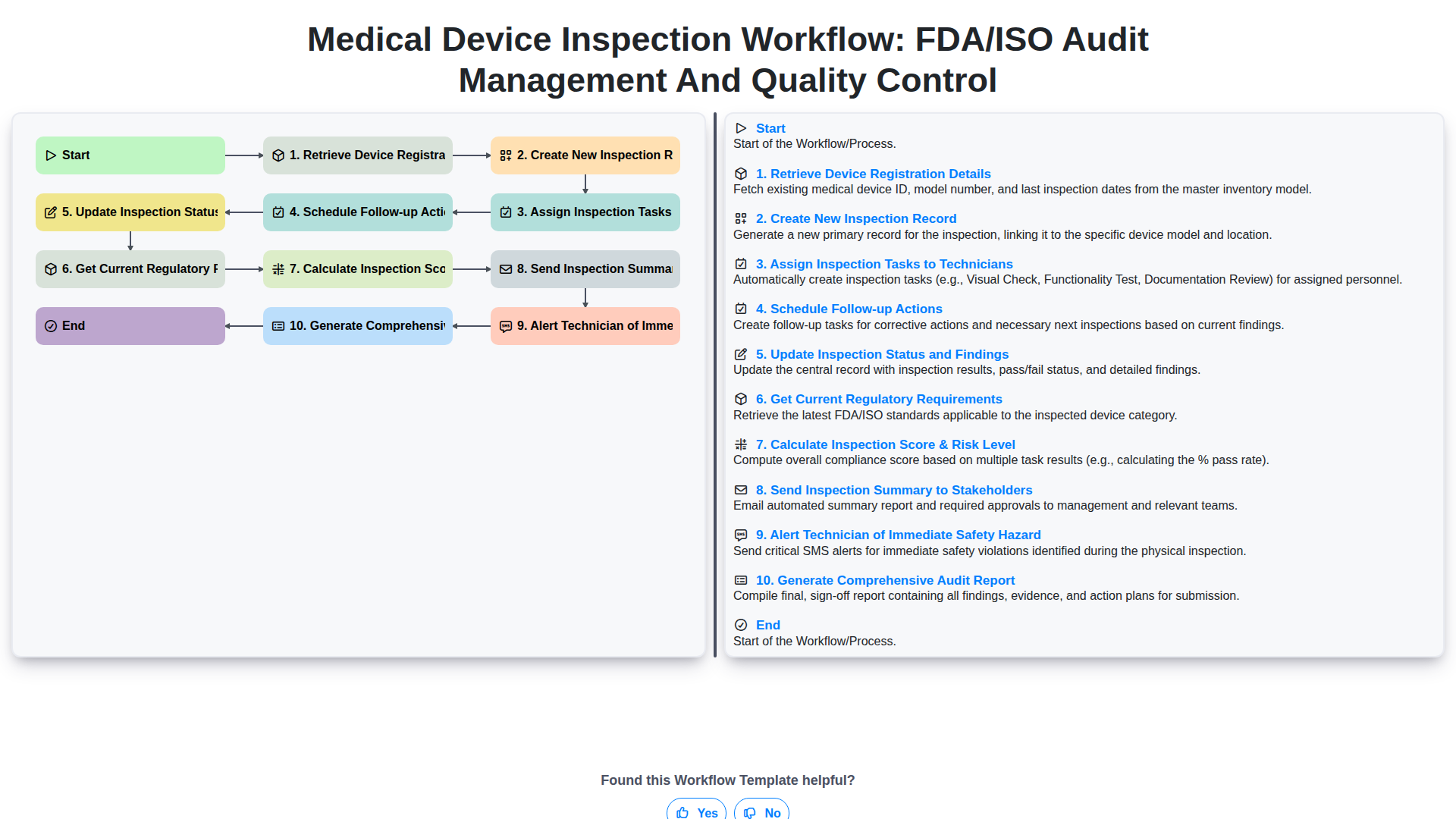
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
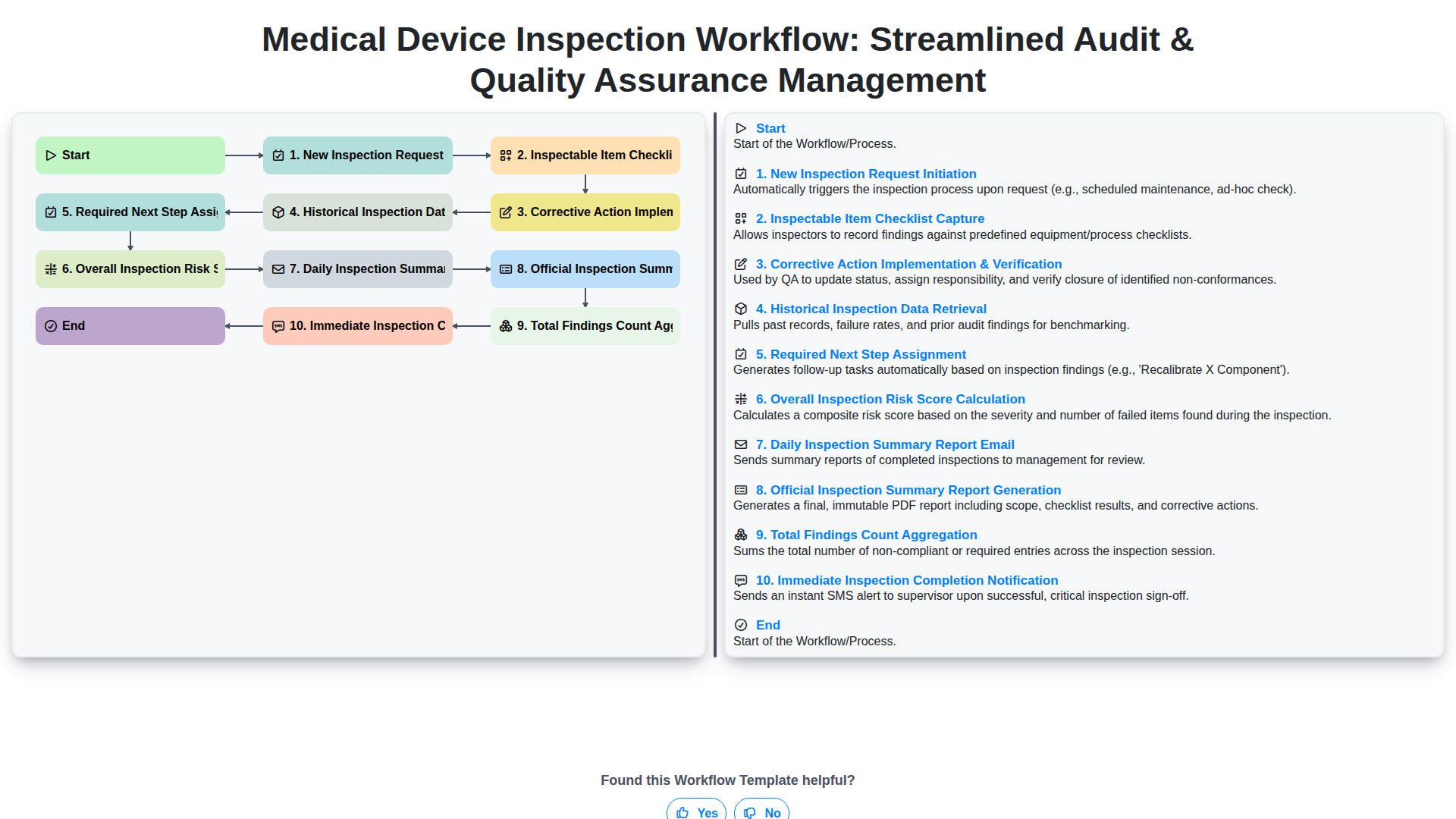
Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
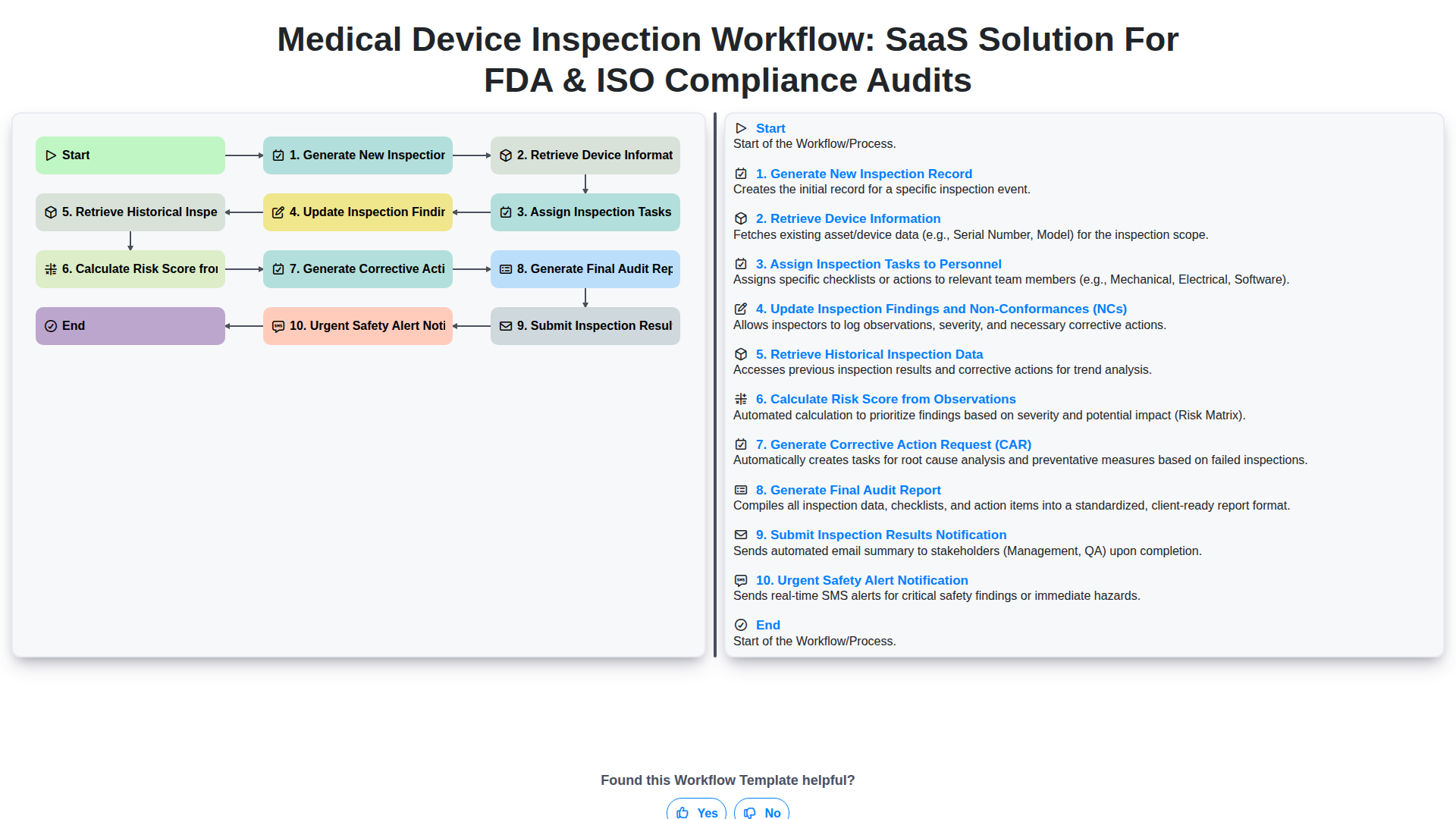
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Streamlined Audit & Compliance Management

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.