Laboratory Equipment Inspection Workflow: GLP/GLP Compliance and Calibration Management
Master Laboratory Equipment Inspection Workflow: Ensure GLP/GLP Compliance with Our Intuitive Inspection Management Solution
This Template was installed 3 times.
Start
Start of the Workflow/Process.
1. Initiate Inspection Task
Create the initial inspection task for the specific piece of laboratory equipment.
2. Retrieve Equipment Serial Number
Get the unique identifier and details for the asset needing inspection (e.g., spectrophotometer, pH meter).
3. Get Associated Calibration Schedule
Retrieve the next due date and required calibration standard from the asset master record.
4. Assign Calibration Task to Technician
Automatically assign the necessary inspection task to the qualified lab technician.
5. Record Inspection Findings
Input inspection results, observations, and readings into the designated data model.
6. Calculate Calibration Due Date
Determine the next service interval based on current date and manufacturer recommendations (e.g., date + 1 year).
7. Generate Calibration Report Draft
Automatically generate the preliminary report structure using collected data.
8. Notify Manager of Task Completion
Send an automated email notification to the Lab Manager upon task completion, attaching preliminary results.
9. Submit Finalized Calibration Record
Submit the final, signed-off record of the calibration and inspection results for record-keeping.
10. Calculate Overall Equipment Health Score
Aggregate multiple inspection data points to generate a composite health score for the equipment.
End
End of the Workflow/Process.
Start of the Workflow/Process.
Create the initial inspection task for the specific piece of laboratory equipment.
Get the unique identifier and details for the asset needing inspection (e.g., spectrophotometer, pH meter).
Retrieve the next due date and required calibration standard from the asset master record.
Automatically assign the necessary inspection task to the qualified lab technician.
Input inspection results, observations, and readings into the designated data model.
Determine the next service interval based on current date and manufacturer recommendations (e.g., date + 1 year).
Automatically generate the preliminary report structure using collected data.
Send an automated email notification to the Lab Manager upon task completion, attaching preliminary results.
Submit the final, signed-off record of the calibration and inspection results for record-keeping.
Aggregate multiple inspection data points to generate a composite health score for the equipment.
End of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
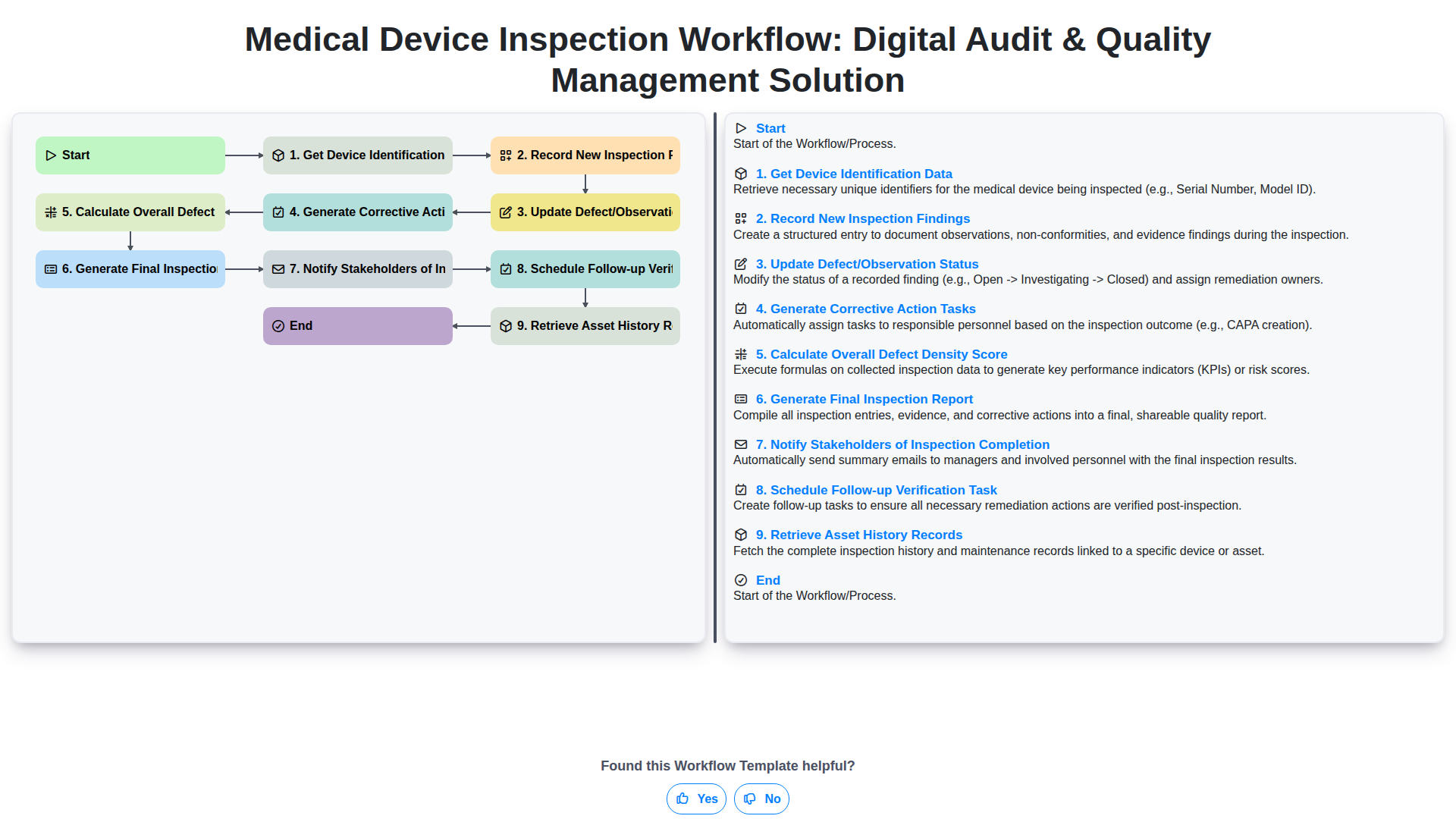
Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
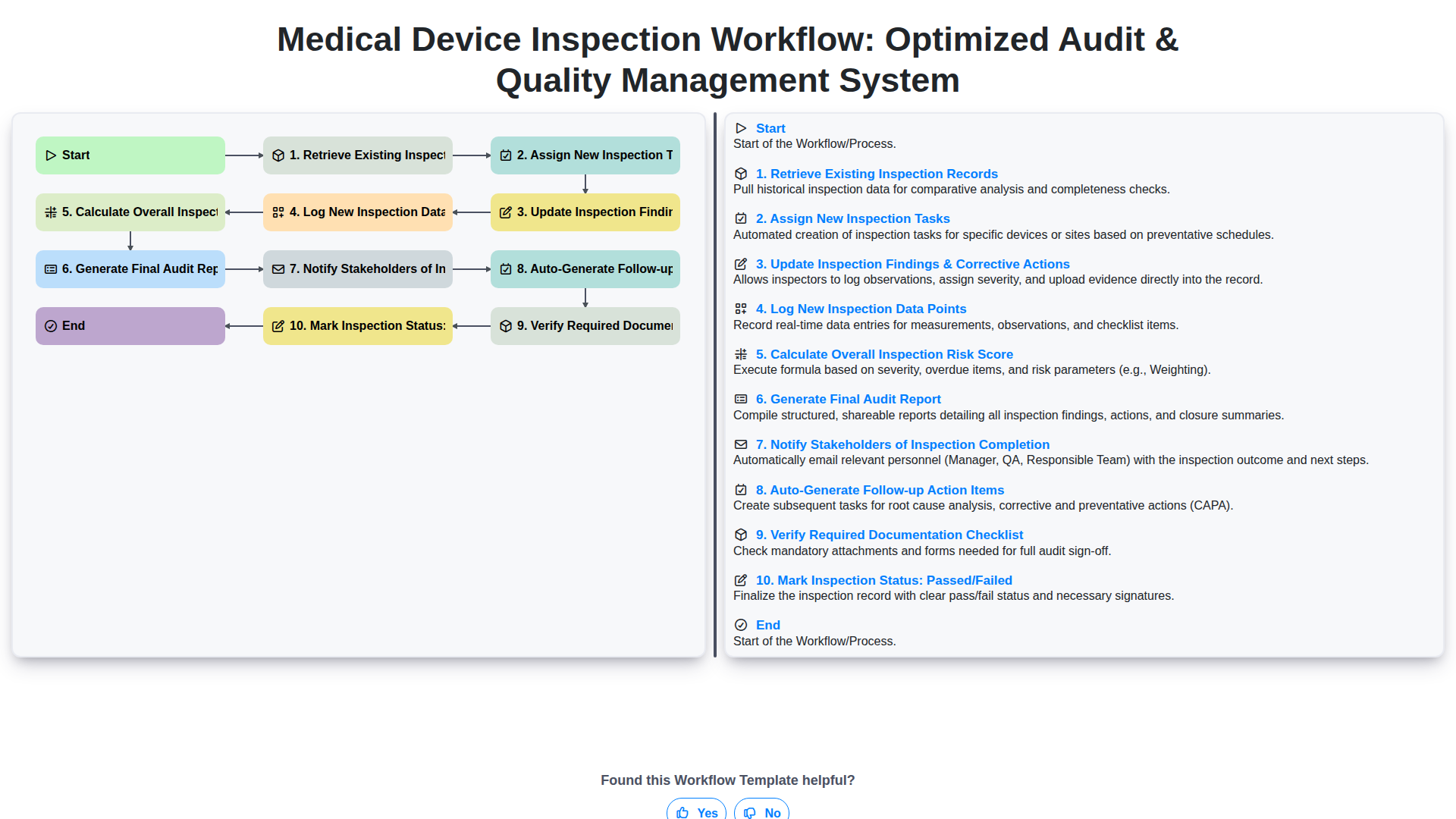
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
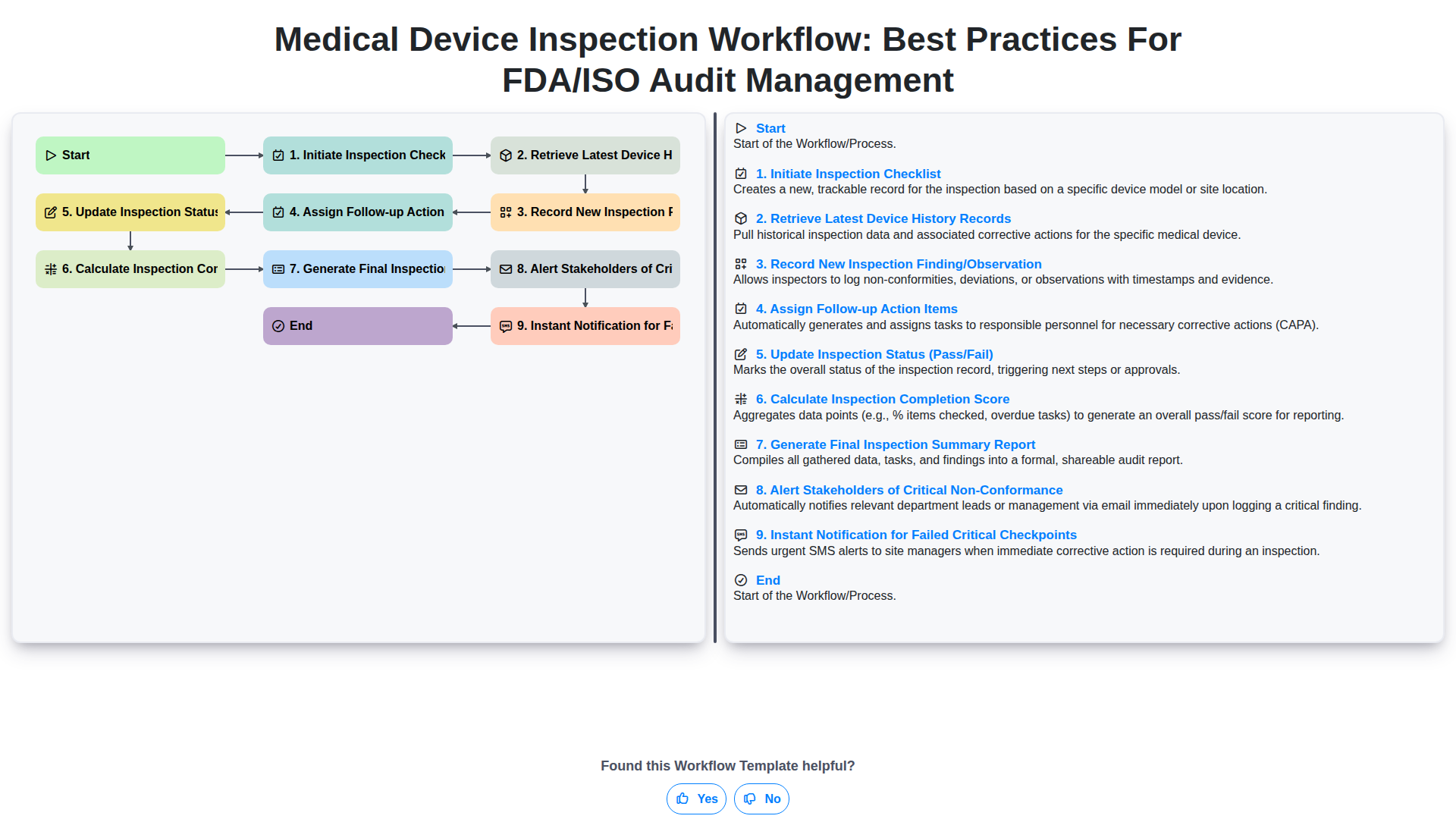
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
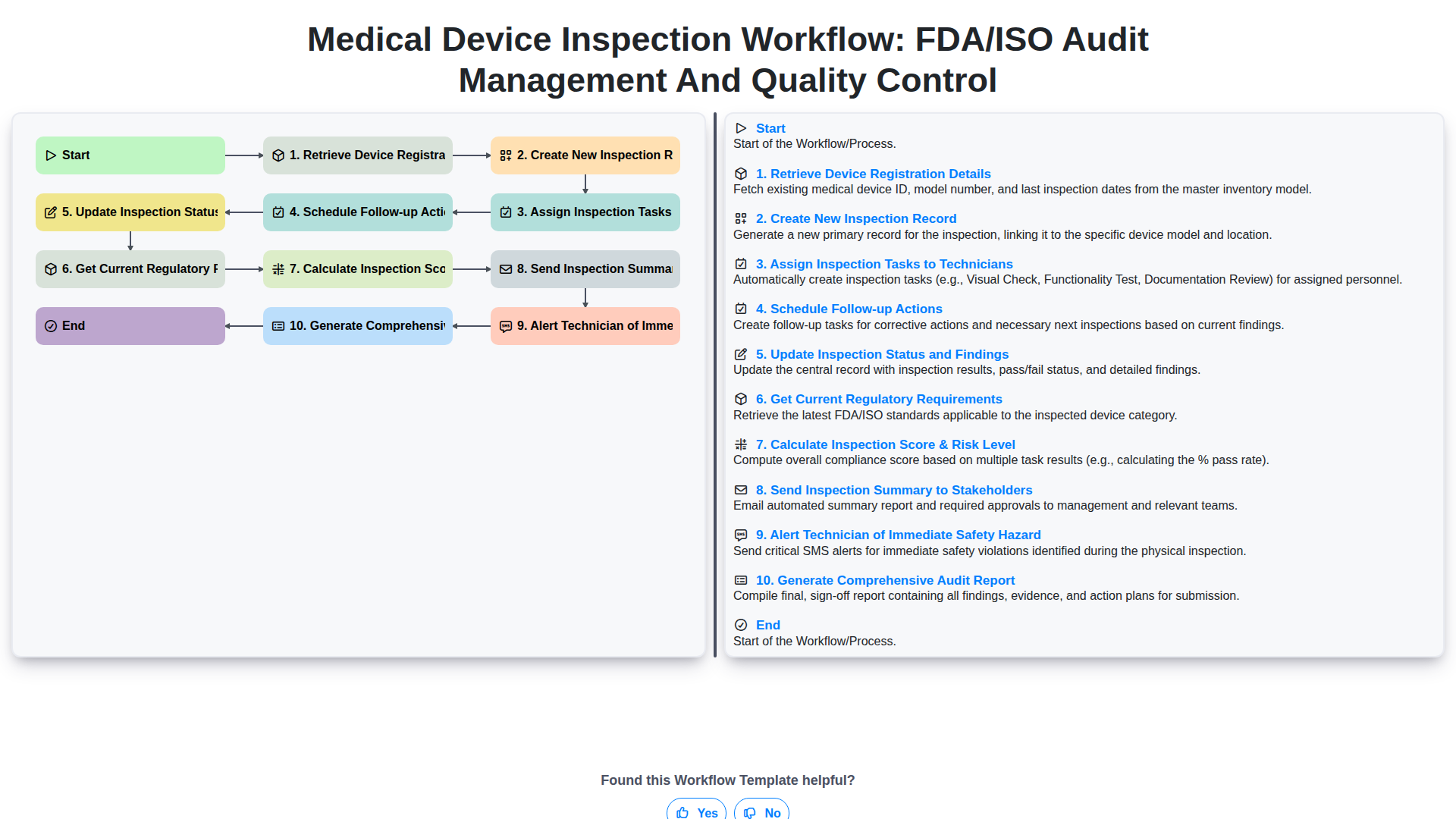
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
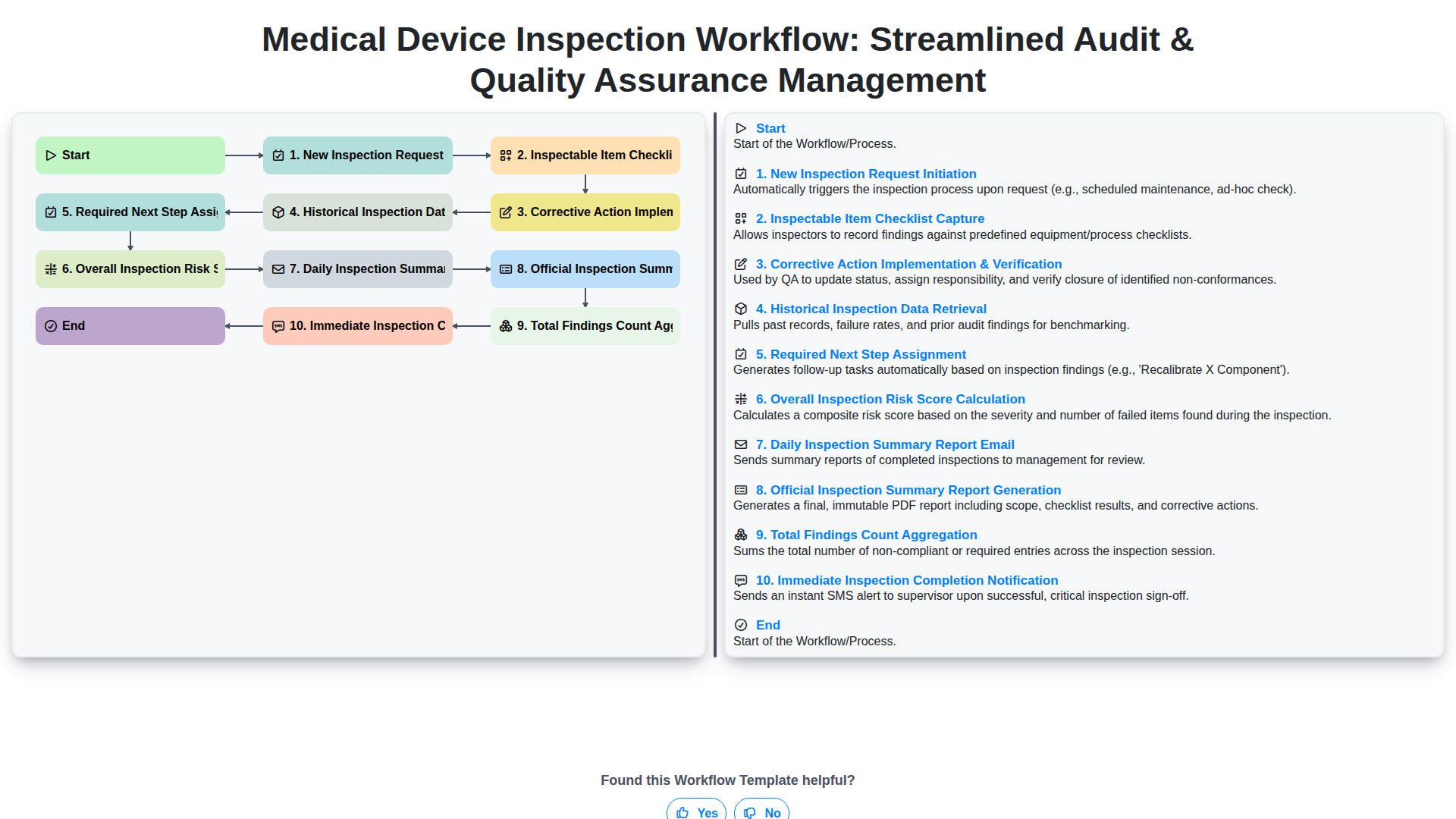
Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
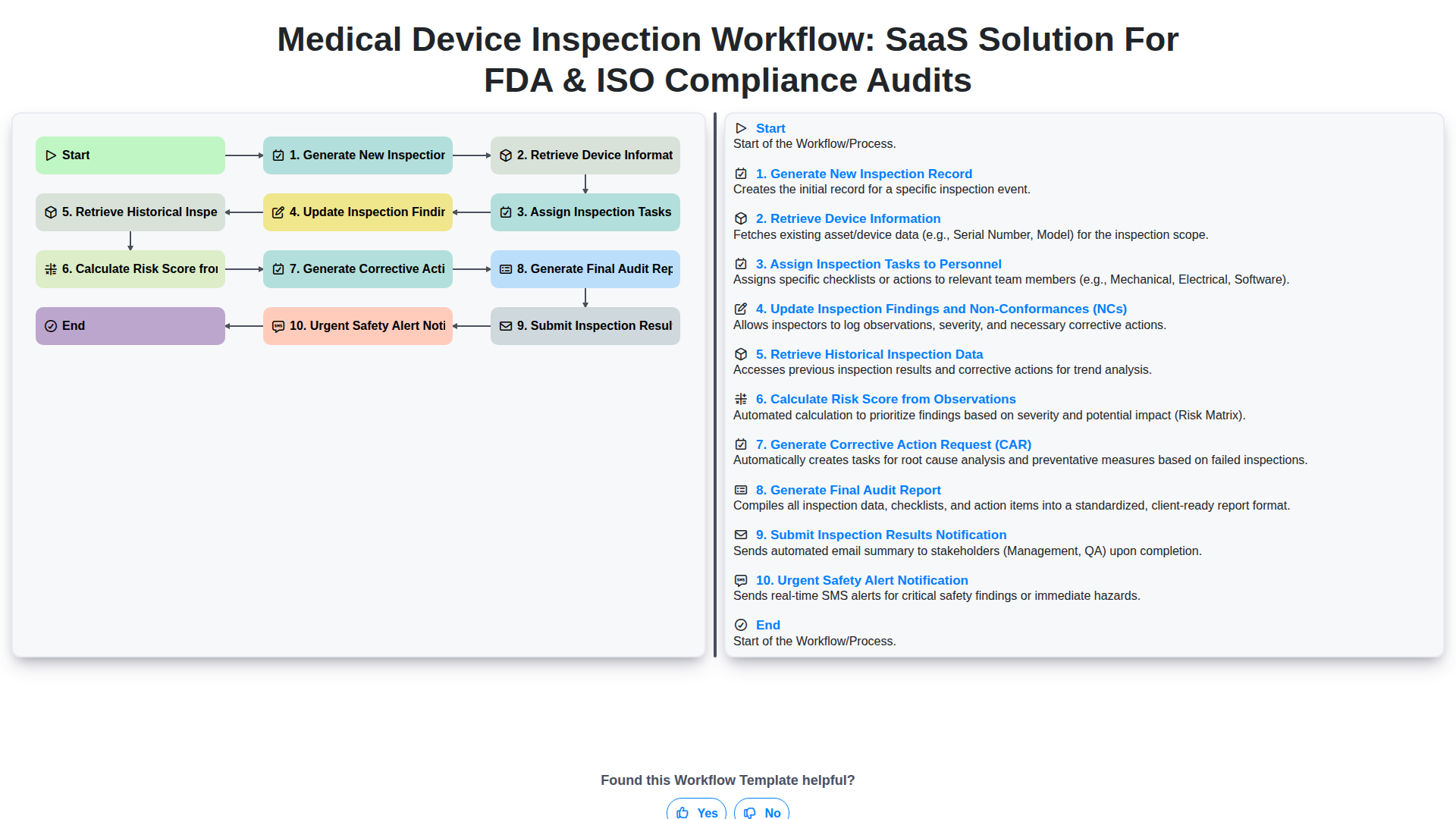
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Streamlined Audit & Compliance Management

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.