Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
Automate Medical Device Inspections: Streamline Audits and Ensure HIPAA/FDA Compliance with Our Workflow Solution
This Template was installed 4 times.
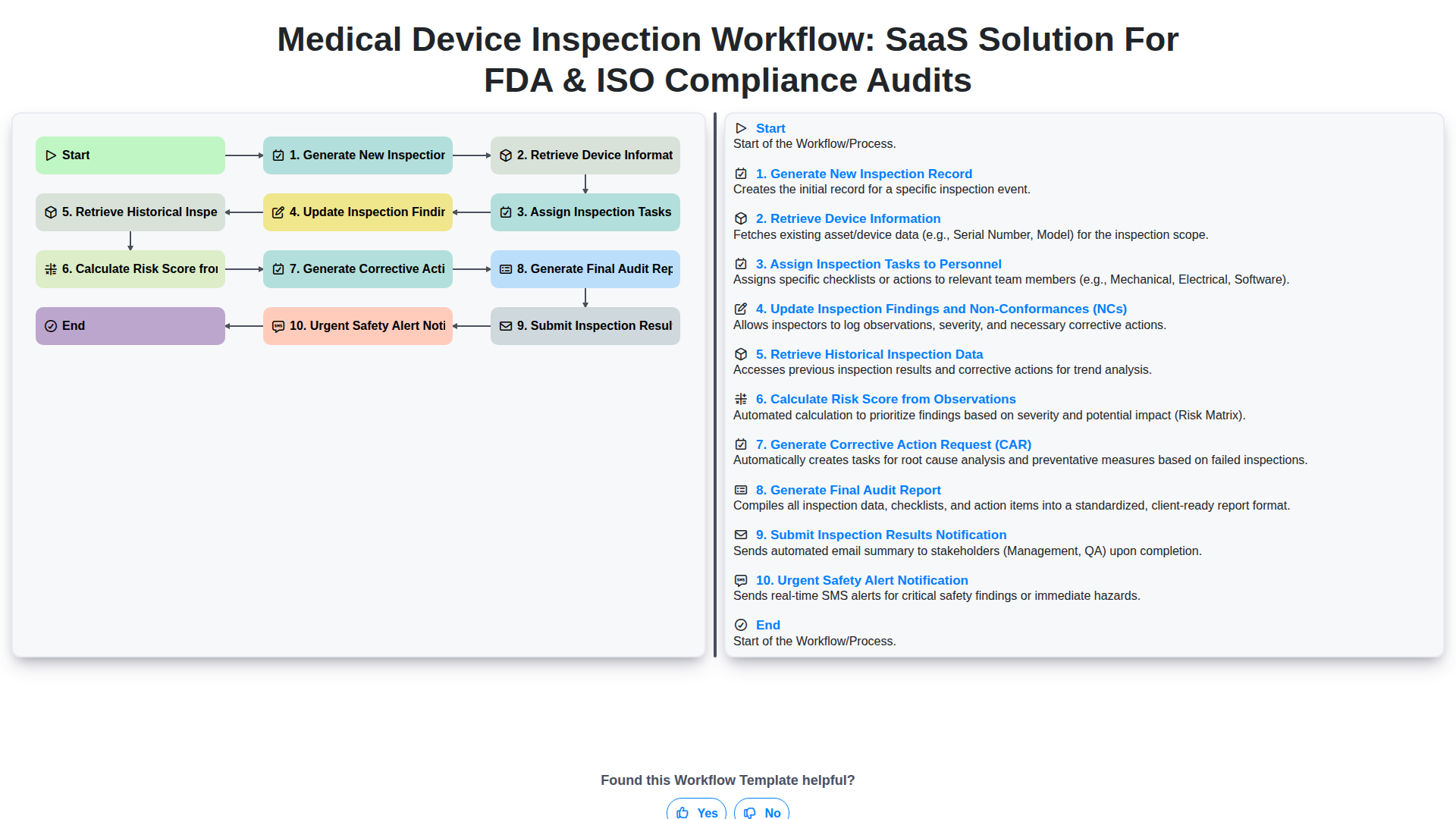
Start
Start of the Workflow/Process.
1. Get Device Identification Data
Retrieve necessary unique identifiers for the medical device being inspected (e.g., Serial Number, Model ID).
2. Record New Inspection Findings
Create a structured entry to document observations, non-conformities, and evidence findings during the inspection.
3. Update Defect/Observation Status
Modify the status of a recorded finding (e.g., Open -> Investigating -> Closed) and assign remediation owners.
4. Generate Corrective Action Tasks
Automatically assign tasks to responsible personnel based on the inspection outcome (e.g., CAPA creation).
5. Calculate Overall Defect Density Score
Execute formulas on collected inspection data to generate key performance indicators (KPIs) or risk scores.
6. Generate Final Inspection Report
Compile all inspection entries, evidence, and corrective actions into a final, shareable quality report.
7. Notify Stakeholders of Inspection Completion
Automatically send summary emails to managers and involved personnel with the final inspection results.
8. Schedule Follow-up Verification Task
Create follow-up tasks to ensure all necessary remediation actions are verified post-inspection.
9. Retrieve Asset History Records
Fetch the complete inspection history and maintenance records linked to a specific device or asset.
End
End of the Workflow/Process.
Start of the Workflow/Process.
Retrieve necessary unique identifiers for the medical device being inspected (e.g., Serial Number, Model ID).
Create a structured entry to document observations, non-conformities, and evidence findings during the inspection.
Modify the status of a recorded finding (e.g., Open -> Investigating -> Closed) and assign remediation owners.
Automatically assign tasks to responsible personnel based on the inspection outcome (e.g., CAPA creation).
Execute formulas on collected inspection data to generate key performance indicators (KPIs) or risk scores.
Compile all inspection entries, evidence, and corrective actions into a final, shareable quality report.
Automatically send summary emails to managers and involved personnel with the final inspection results.
Create follow-up tasks to ensure all necessary remediation actions are verified post-inspection.
Fetch the complete inspection history and maintenance records linked to a specific device or asset.
End of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
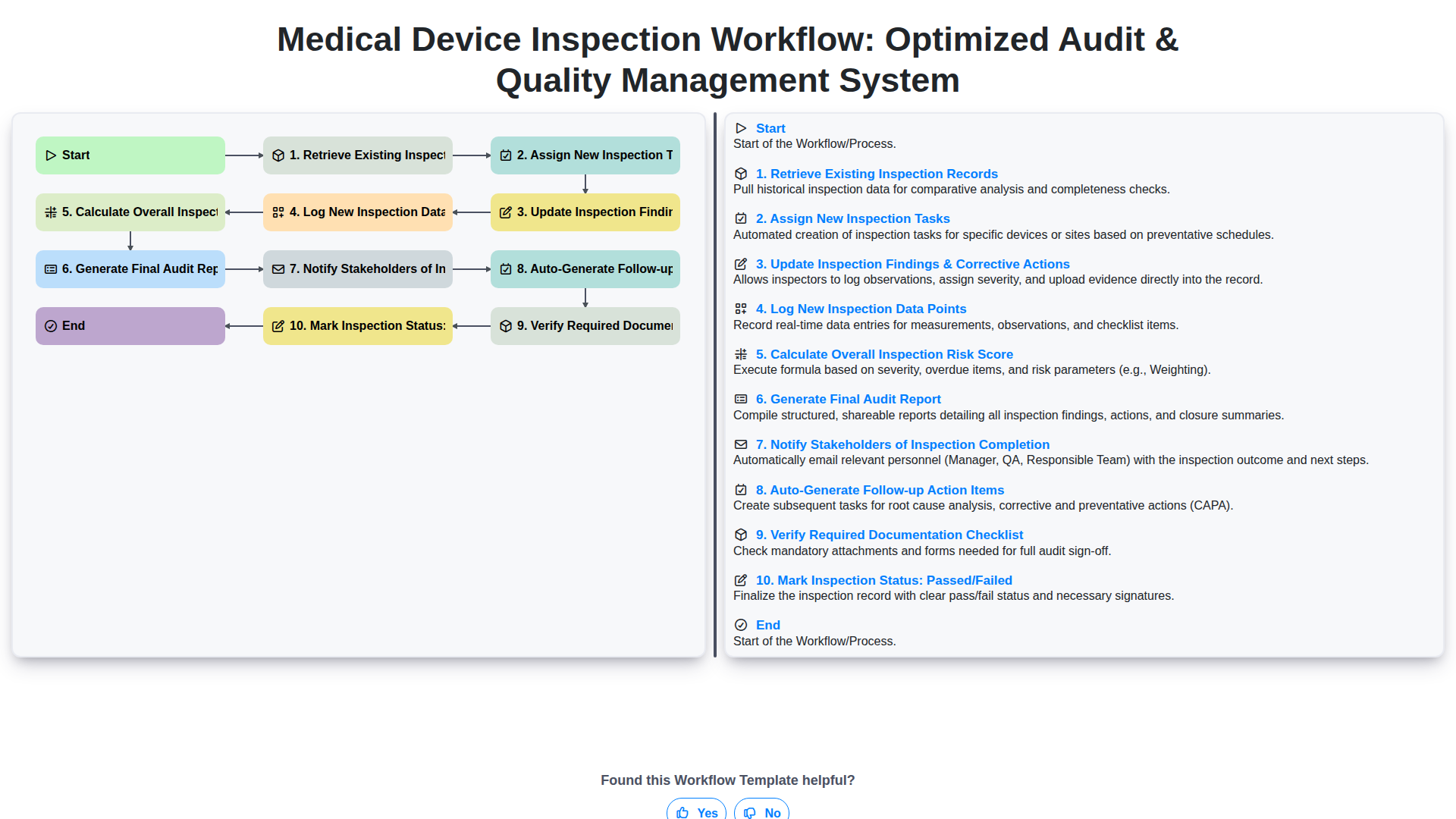
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
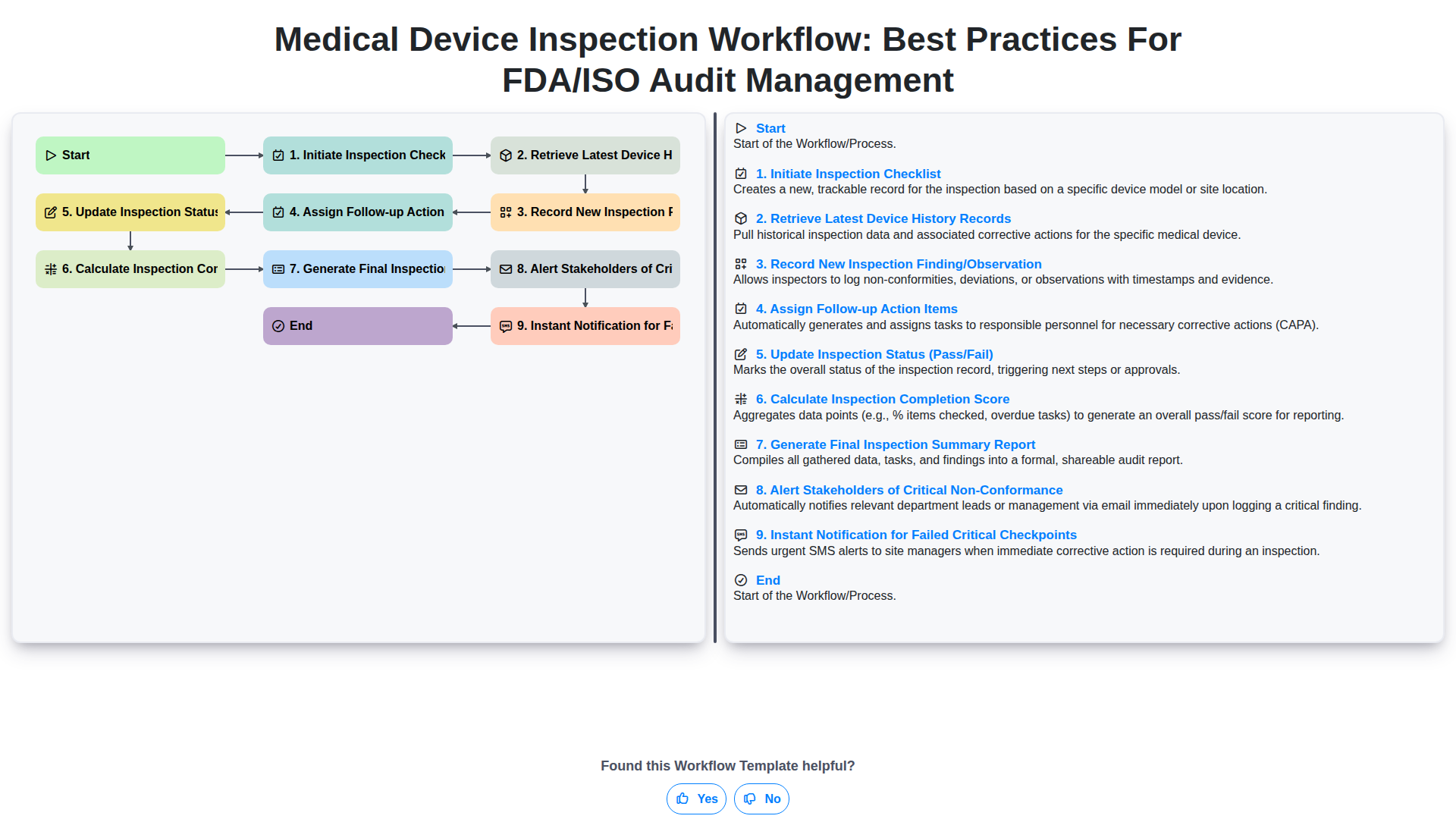
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
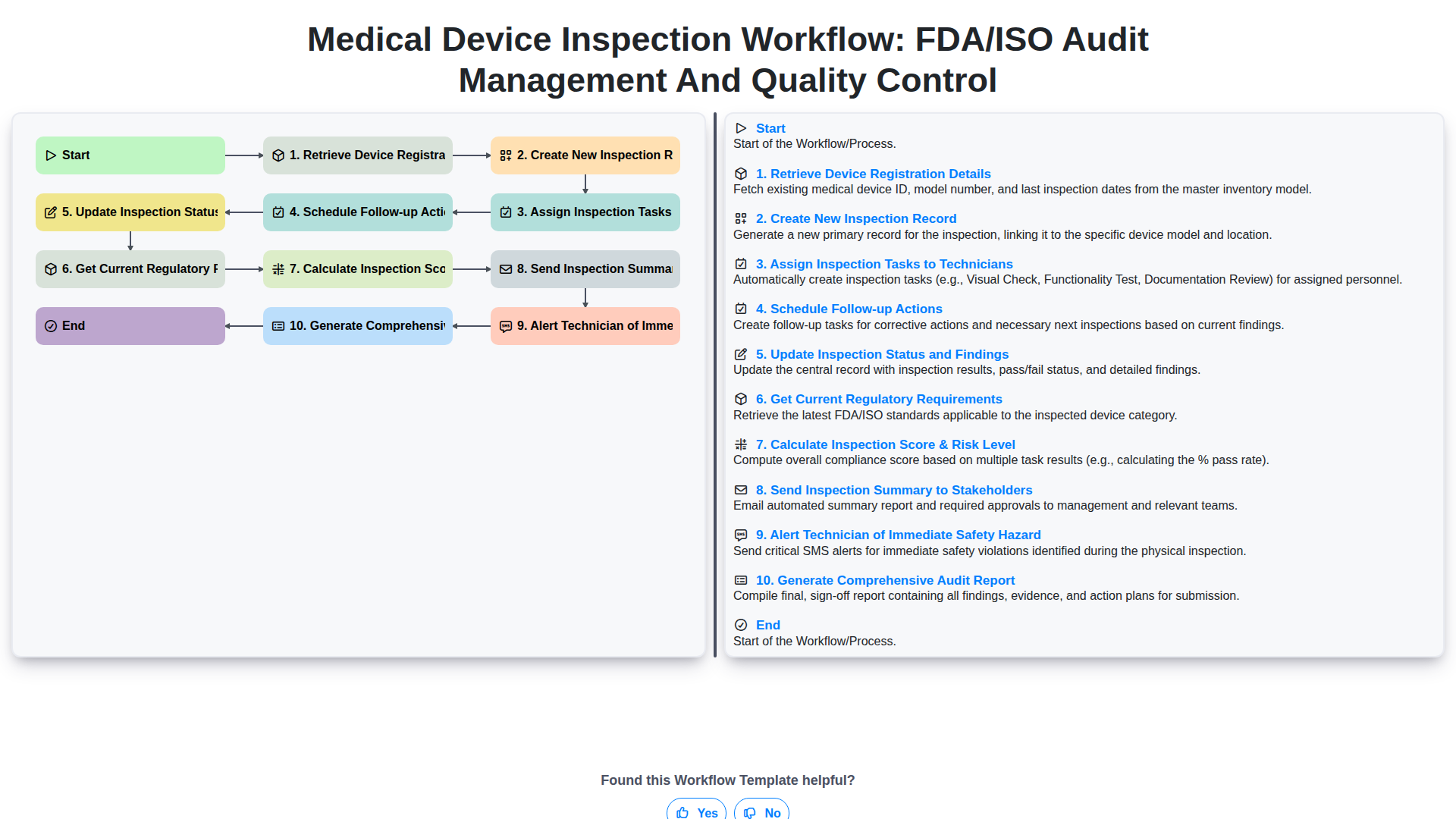
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
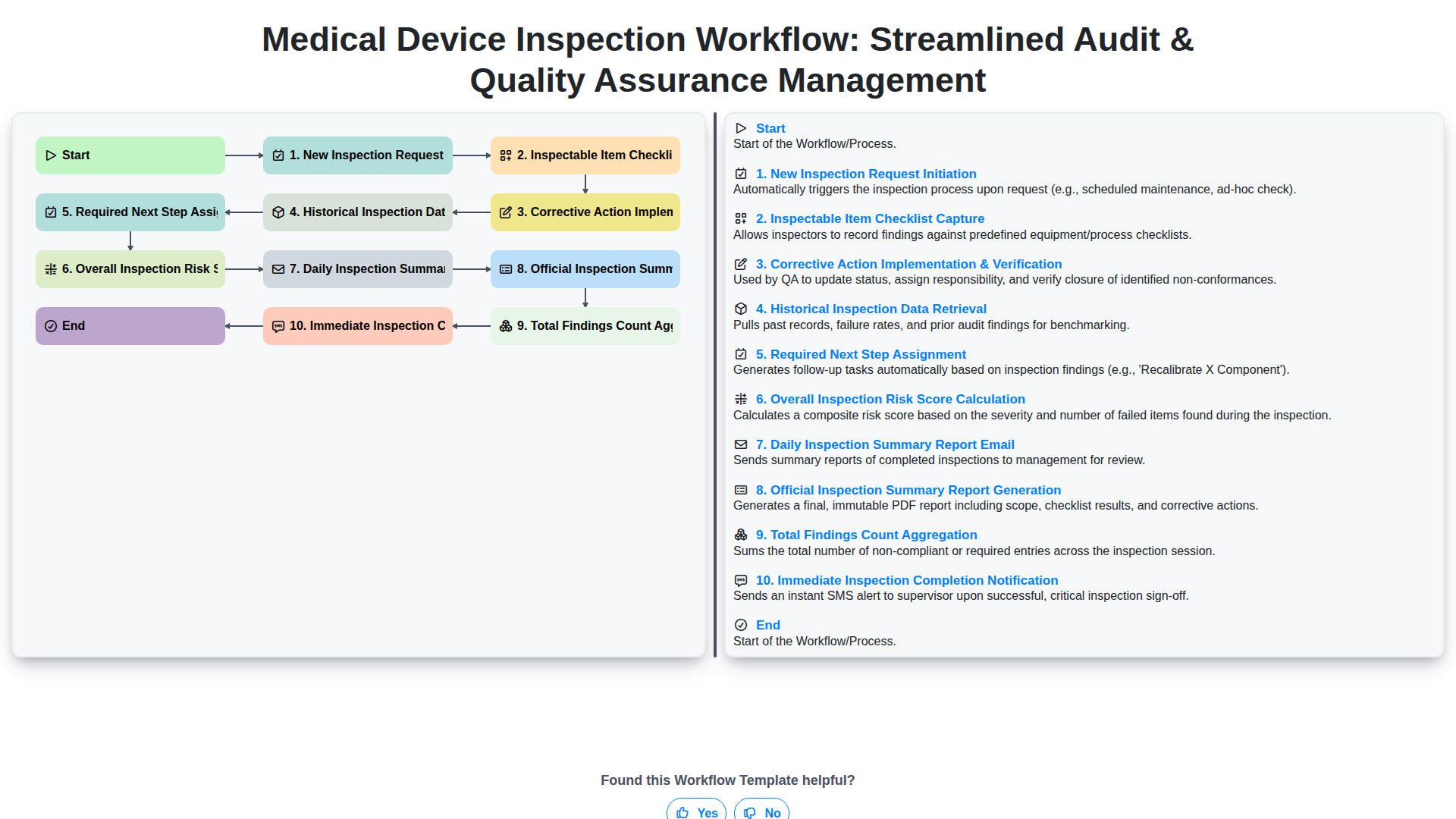
Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Streamlined Audit & Compliance Management

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
Medical Device Inspection Workflow: FDA & Quality Compliance Tracking
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.