Medical Device Inspection Workflow: FDA & Quality Compliance Tracking
Streamline Your Medical Device Inspections: Achieve FDA & Quality Compliance with Our Automated Inspection Management Workflow. Effortlessly track, document, and manage all steps of your medical device inspections for effortless regulatory adherence.
This Template was installed 4 times.
Start
Start of the Workflow/Process.
1. Initial Inspection Task Assignment
Creates the initial inspection task for the assigned team member based on facility location or device type.
2. Retrieve Equipment Details
Fetches specific details (e.g., Serial Number, Model, Calibration Date) from the Equipment Inventory Data Model.
3. Record Inspection Findings
Allows inspectors to document observations, pass/fail status, and corrective actions for specific inspection checklist items.
4. Generate Deficiency Report Entry
Creates a formal record of non-conformances found during the inspection process.
5. Calculate Risk Score
Executes a formula to determine the overall risk level based on aggregated findings (e.g., assigning weights to severity).
6. Automated Follow-up Task Generation
Creates subsequent tasks for necessary follow-up actions based on critical findings (e.g., 'Immediate Remediation Required').
7. Submit Inspection Summary Email
Sends a summary report via email to Quality Assurance and Management upon completion.
8. Generate Final Inspection Report
Compiles all data (findings, actions, status) into a final, signed-off, auditable PDF report.
9. Aggregate Inspection Results
Aggregates all findings to calculate overall pass/fail metrics and trend analysis.
10. Schedule Corrective Action Meeting
Automatically schedules a review meeting task for stakeholders to discuss necessary remediation steps.
End
End of the Workflow/Process.
Start of the Workflow/Process.
Creates the initial inspection task for the assigned team member based on facility location or device type.
Fetches specific details (e.g., Serial Number, Model, Calibration Date) from the Equipment Inventory Data Model.
Allows inspectors to document observations, pass/fail status, and corrective actions for specific inspection checklist items.
Creates a formal record of non-conformances found during the inspection process.
Executes a formula to determine the overall risk level based on aggregated findings (e.g., assigning weights to severity).
Creates subsequent tasks for necessary follow-up actions based on critical findings (e.g., 'Immediate Remediation Required').
Sends a summary report via email to Quality Assurance and Management upon completion.
Compiles all data (findings, actions, status) into a final, signed-off, auditable PDF report.
Aggregates all findings to calculate overall pass/fail metrics and trend analysis.
Automatically schedules a review meeting task for stakeholders to discuss necessary remediation steps.
End of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
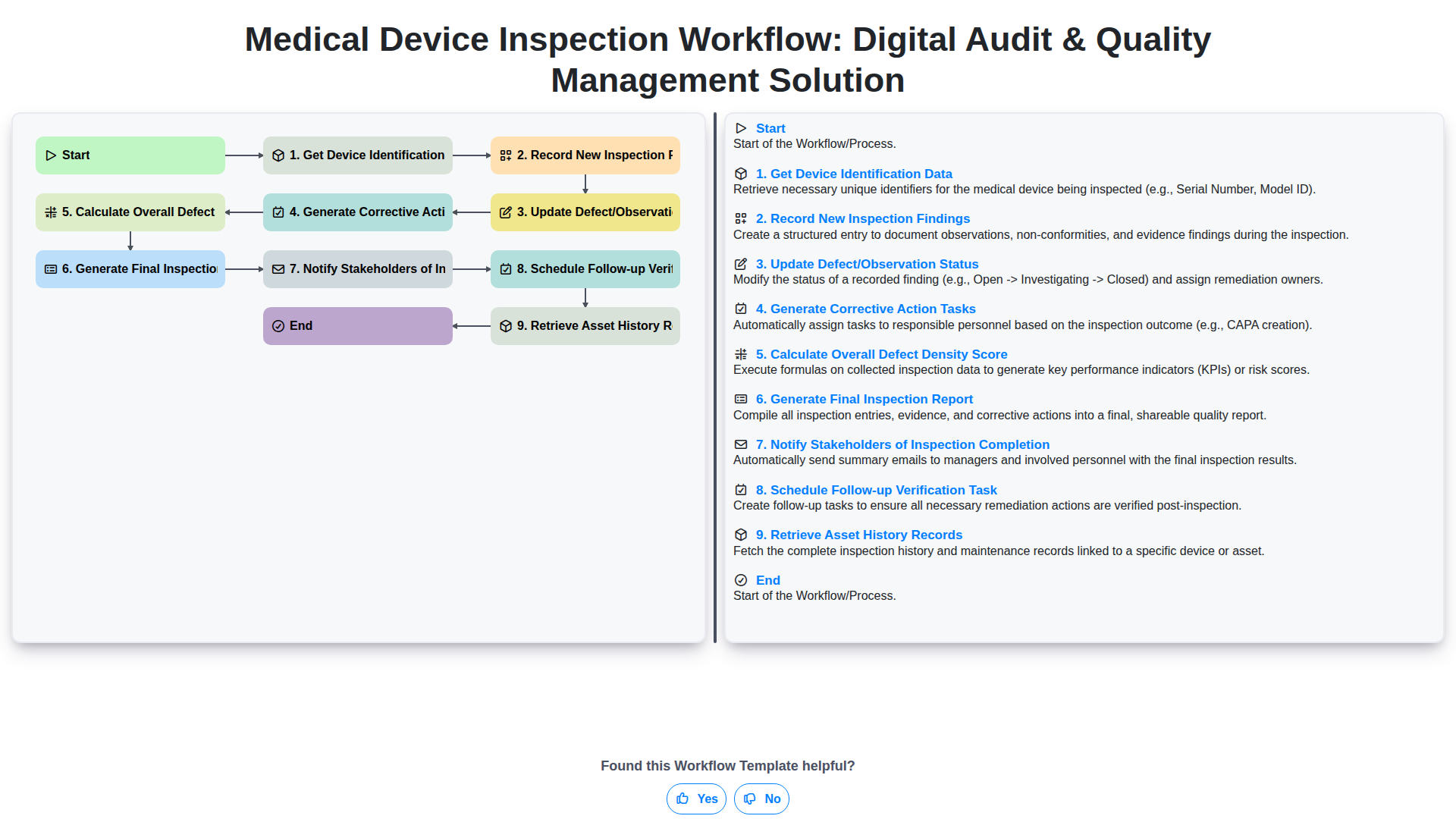
Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
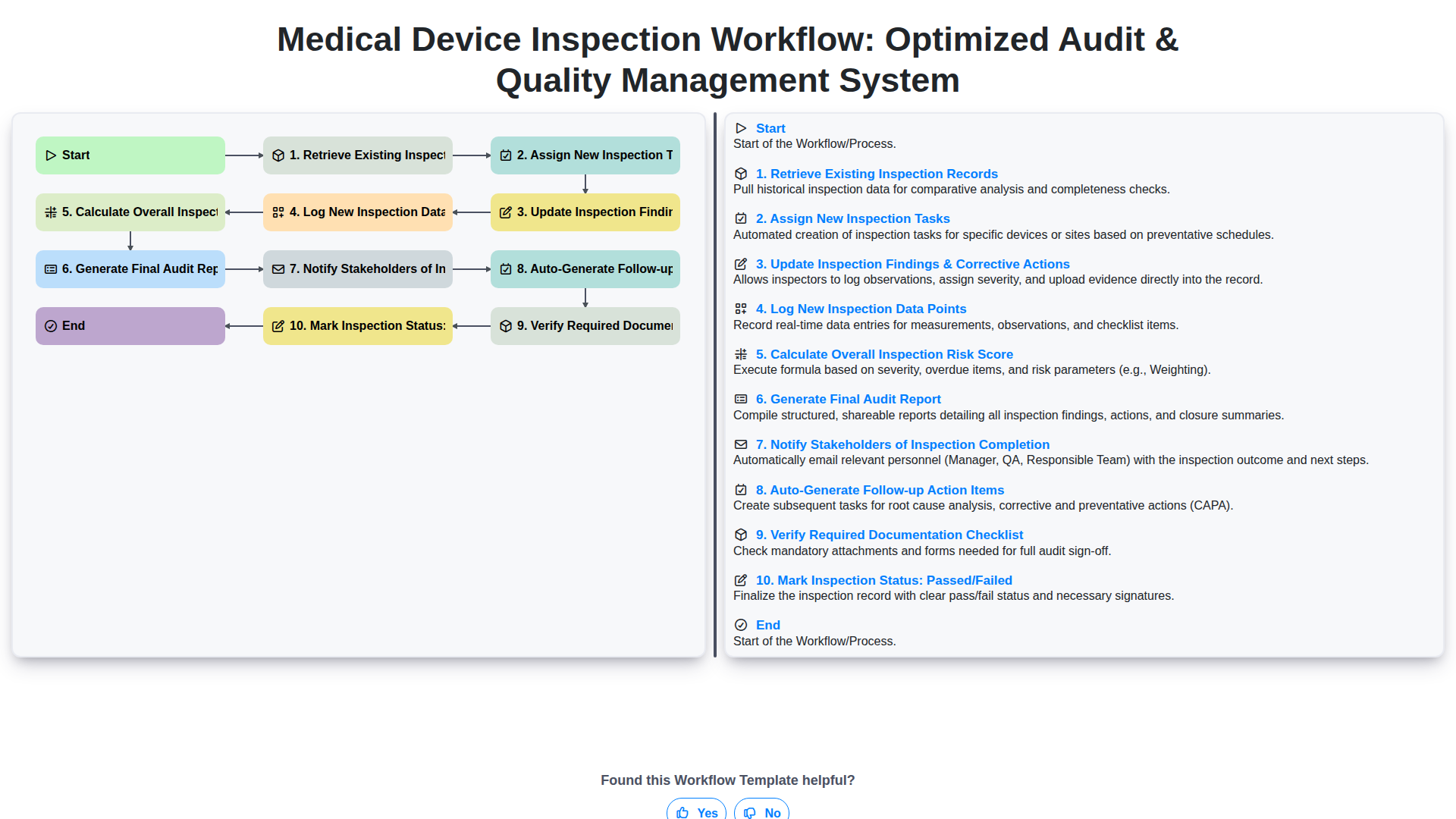
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
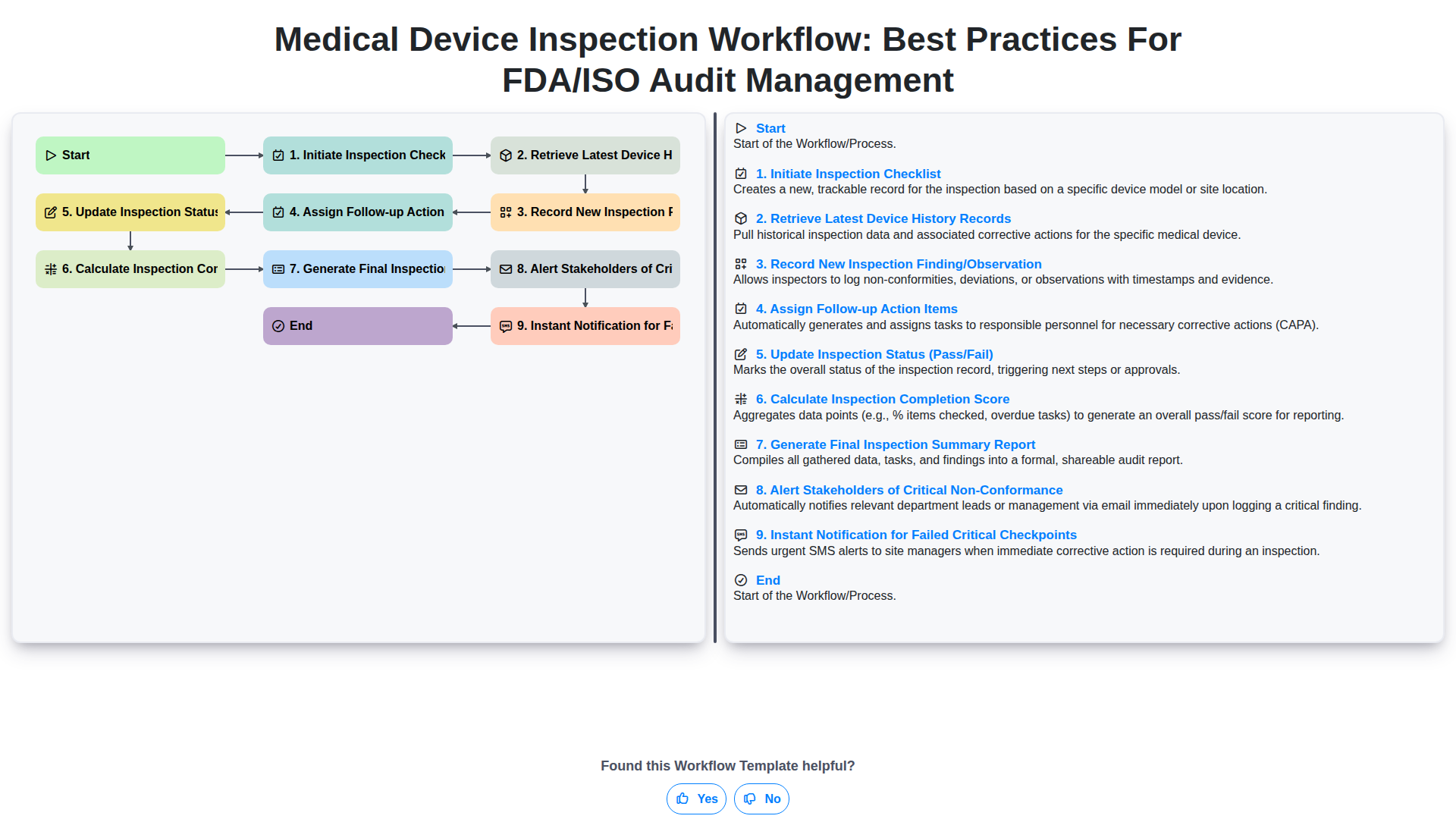
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
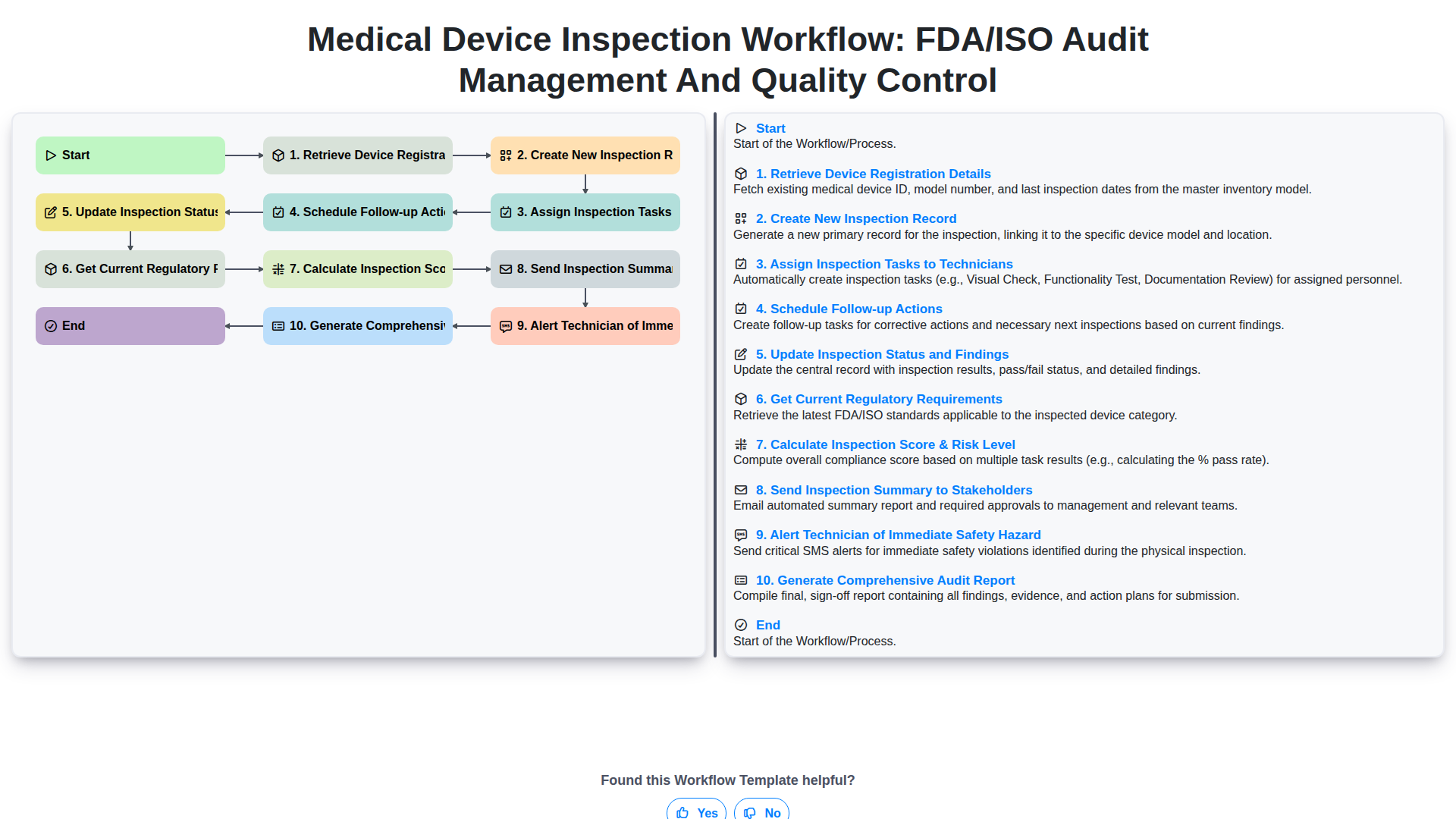
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
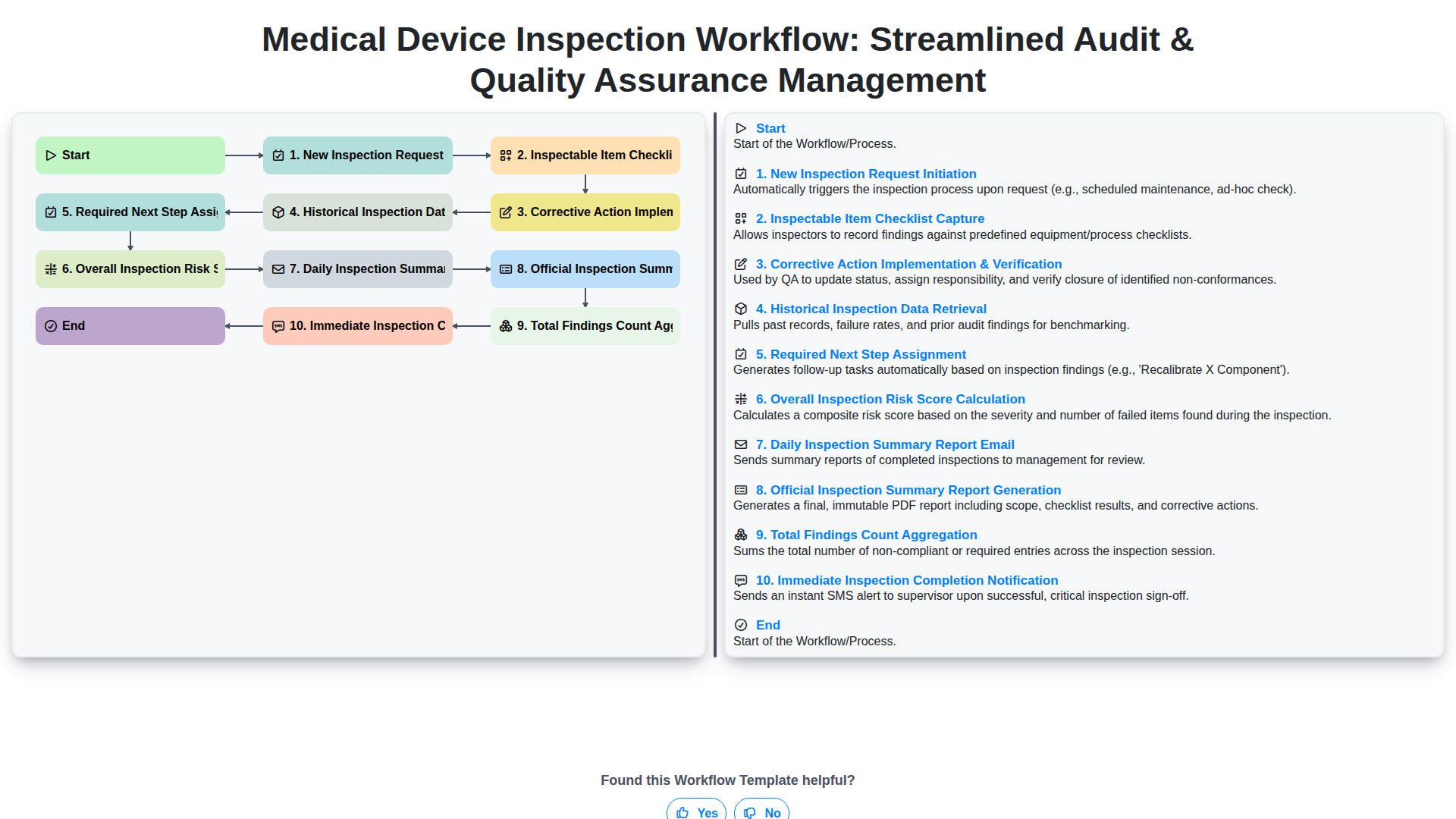
Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
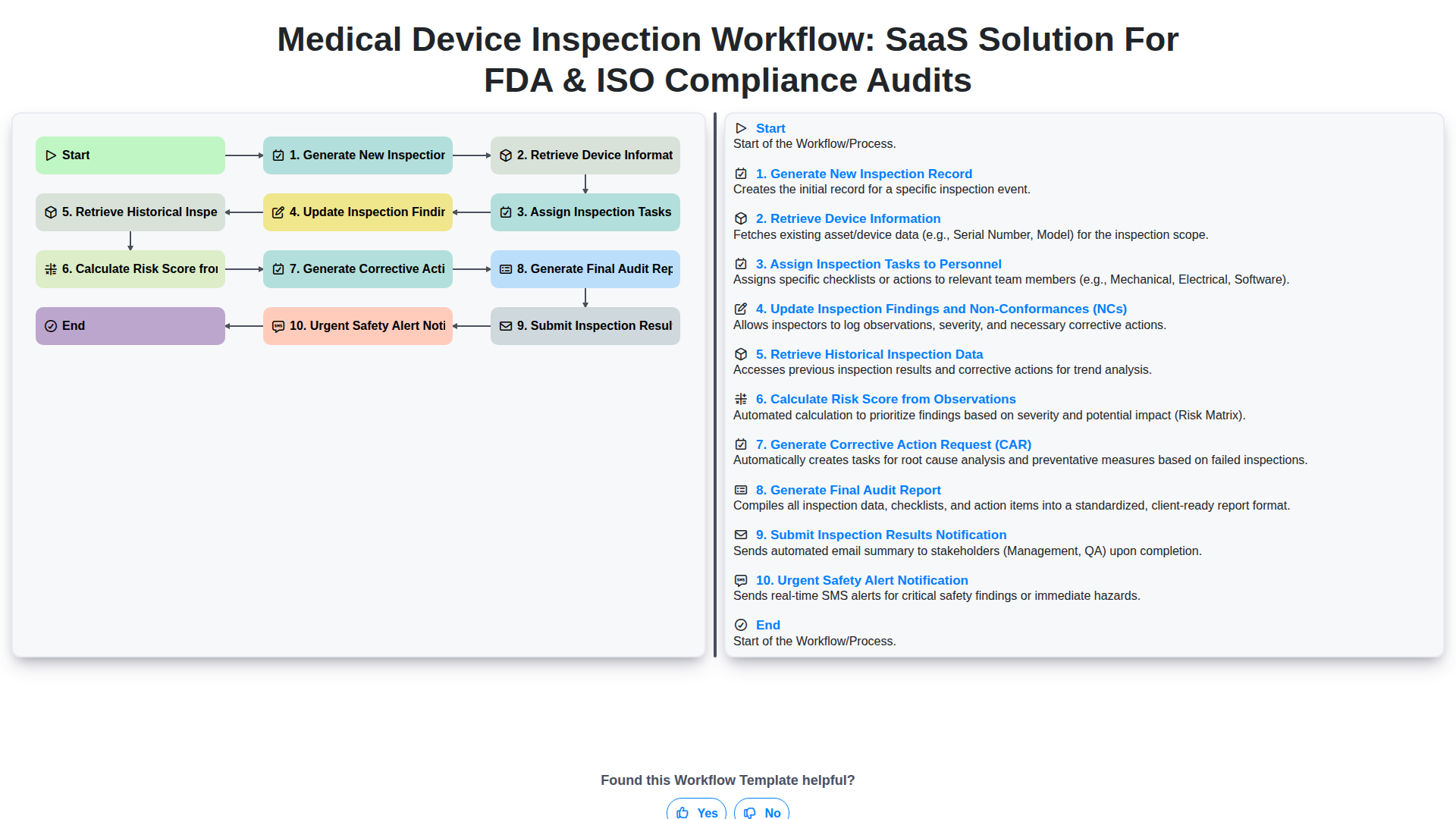
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Streamlined Audit & Compliance Management

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.