Medical Device Inspection Workflow: Streamlined Audit & Compliance Management
Automate Medical Device Inspections: Simplify Audits, Ensure FDA Compliance, and Accelerate Quality Sign-Offs with Our Workflow Solution
This Template was installed 3 times.
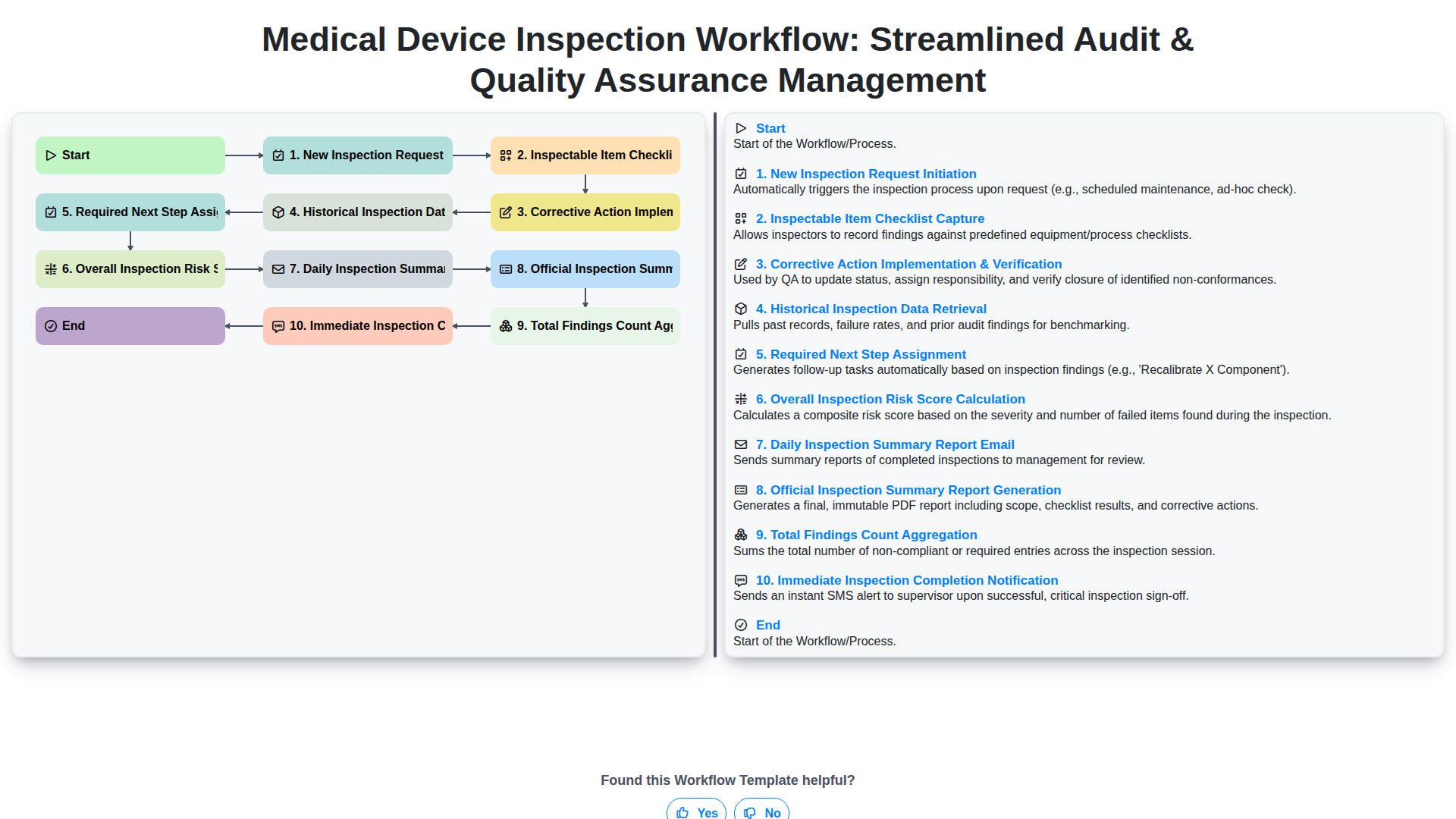
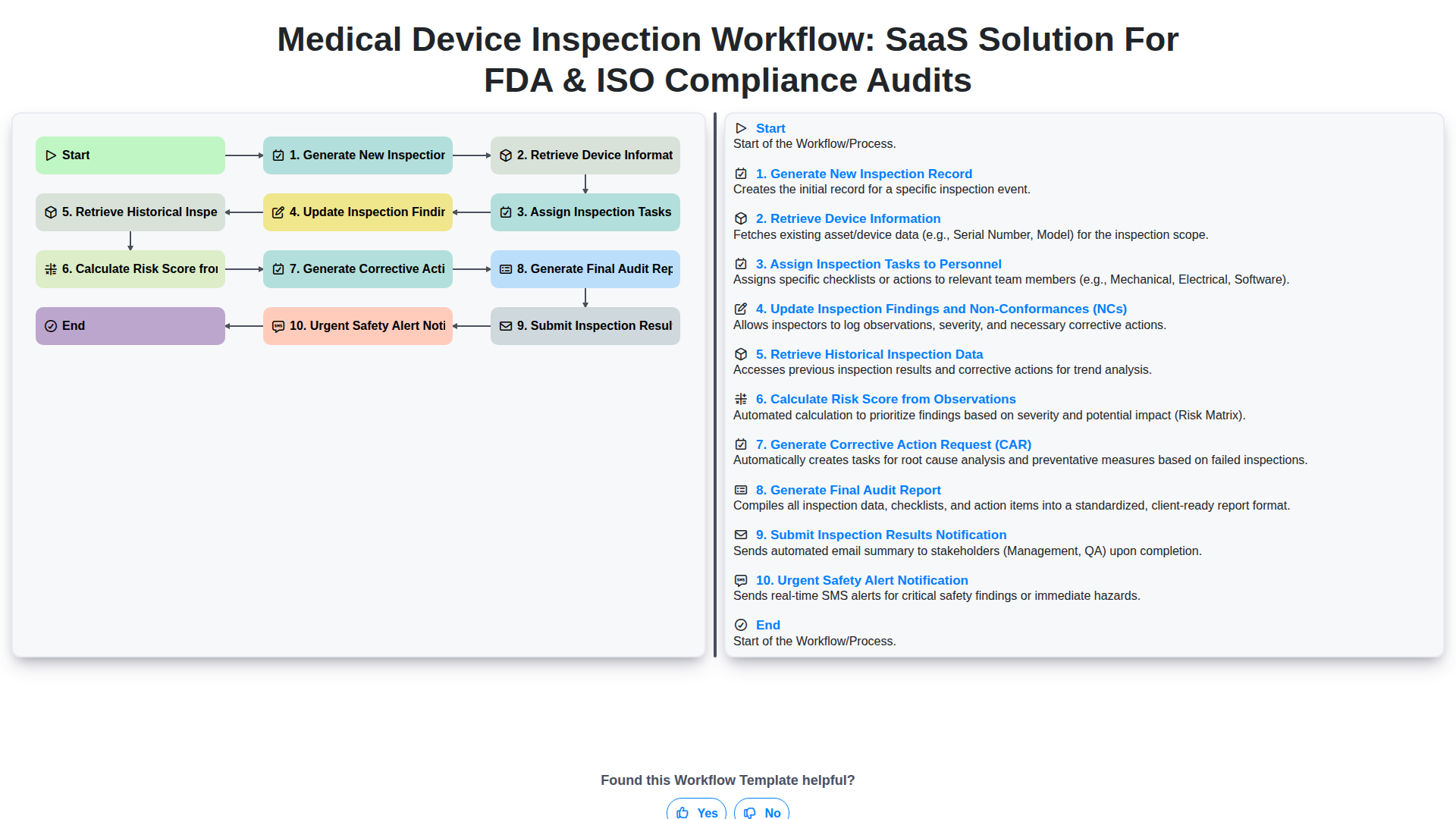
Start
Start of the Workflow/Process.
1. Initialize Inspection Record
Creates a new inspection record, capturing essential details like inspection date, device ID, and assigned inspector.
2. Retrieve Device Master Data
Fetches necessary background information (e.g., Model Number, Calibration Schedule) for the specific medical device being inspected.
3. Generate Task List & Assign Inspectors
Automatically generates the required inspection checklist tasks and assigns them to relevant team members.
4. Perform On-Site Inspection Steps
Guides the inspector through standardized, sequential physical inspection steps using interactive checklists.
5. Document Inspection Findings & Observations
Allows inspectors to record detailed observations, including pass/fail status, non-conformances, and corrective actions directly on the device record.
6. Update Non-Conformance Details
Updates the severity, root cause, and proposed corrective actions for identified defects or non-conformances.
7. Calculate Overall Risk Score
Executes a formula based on collected data (e.g., weighted average of failure points) to determine the overall risk level of the device.
8. Review and Approve Inspection Report
Creates a final review task assigned to the Quality Manager for official sign-off.
9. Generate Final Inspection Report
Compiles all collected data, tasks completed, and identified non-conformances into a comprehensive, traceable final report.
10. Notify Stakeholders of Completion
Automatically emails the final report and next steps to relevant parties (e.g., Engineering, Quality, Management).
11. Schedule Follow-up Actions
Generates follow-up tasks and due dates for necessary Corrective and Preventive Actions (CAPA).
End
End of the Workflow/Process.
Start of the Workflow/Process.
Creates a new inspection record, capturing essential details like inspection date, device ID, and assigned inspector.
Fetches necessary background information (e.g., Model Number, Calibration Schedule) for the specific medical device being inspected.
Automatically generates the required inspection checklist tasks and assigns them to relevant team members.
Guides the inspector through standardized, sequential physical inspection steps using interactive checklists.
Allows inspectors to record detailed observations, including pass/fail status, non-conformances, and corrective actions directly on the device record.
Updates the severity, root cause, and proposed corrective actions for identified defects or non-conformances.
Executes a formula based on collected data (e.g., weighted average of failure points) to determine the overall risk level of the device.
Creates a final review task assigned to the Quality Manager for official sign-off.
Compiles all collected data, tasks completed, and identified non-conformances into a comprehensive, traceable final report.
Automatically emails the final report and next steps to relevant parties (e.g., Engineering, Quality, Management).
Generates follow-up tasks and due dates for necessary Corrective and Preventive Actions (CAPA).
End of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
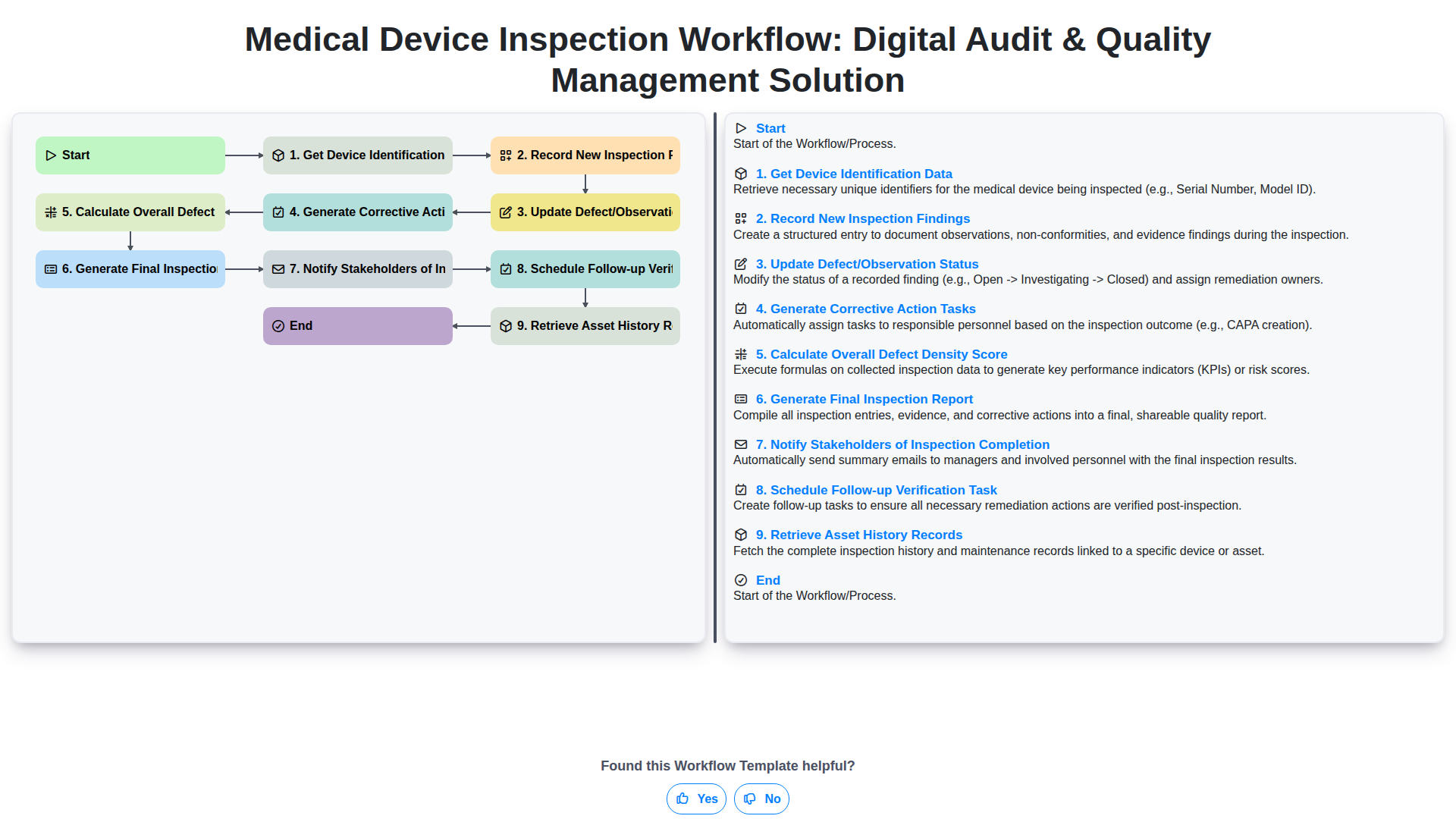
Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
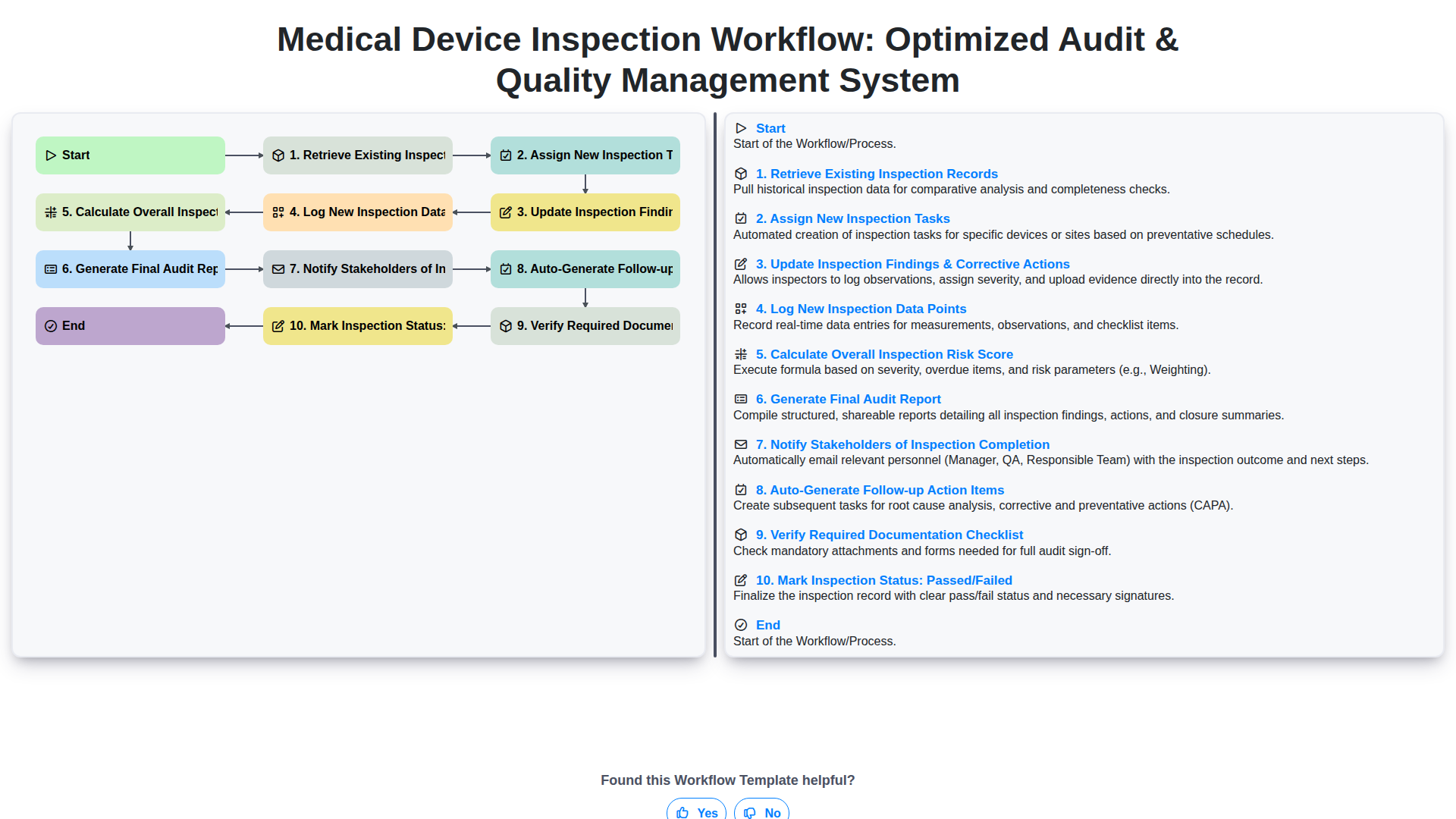
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
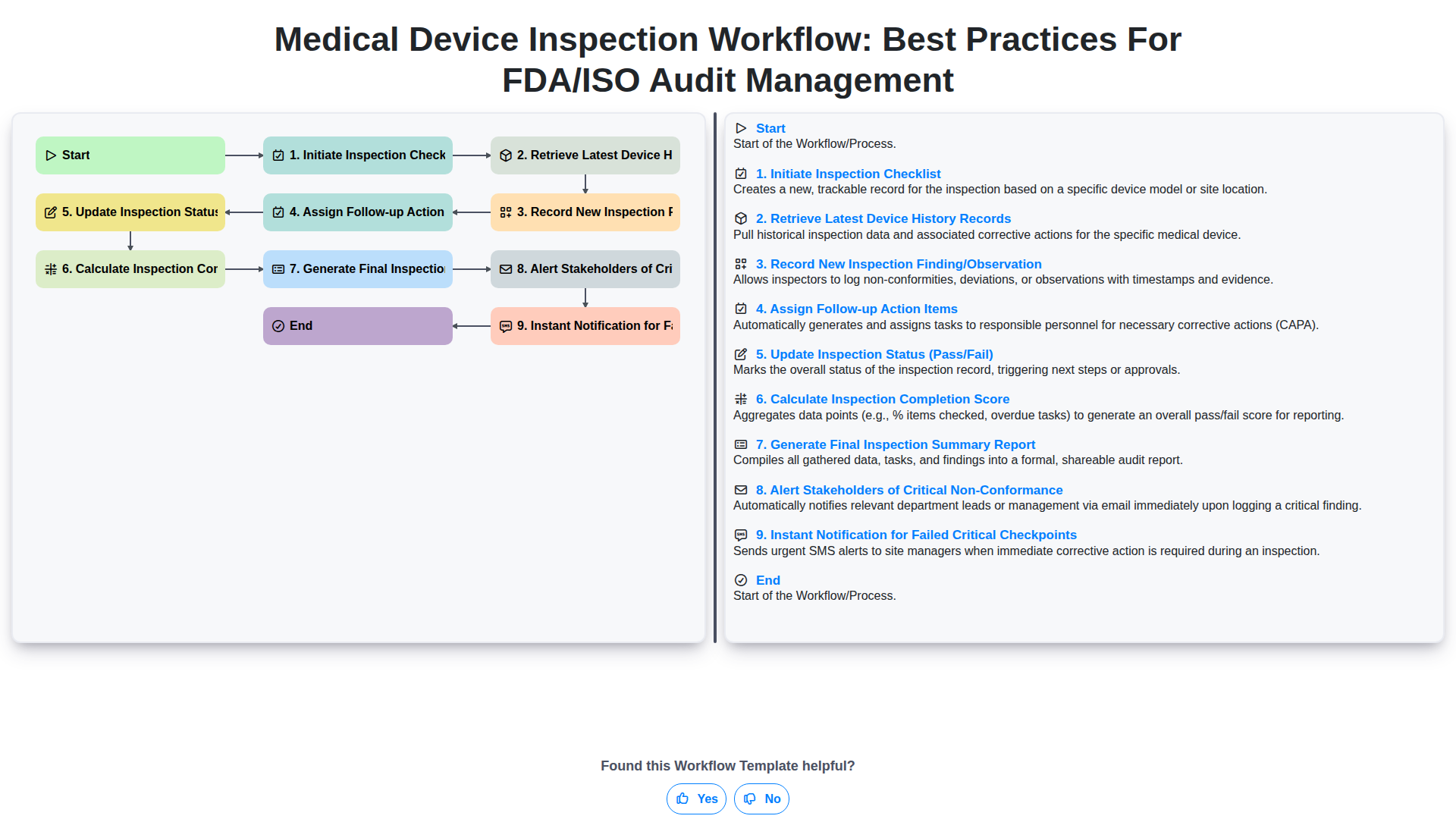
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
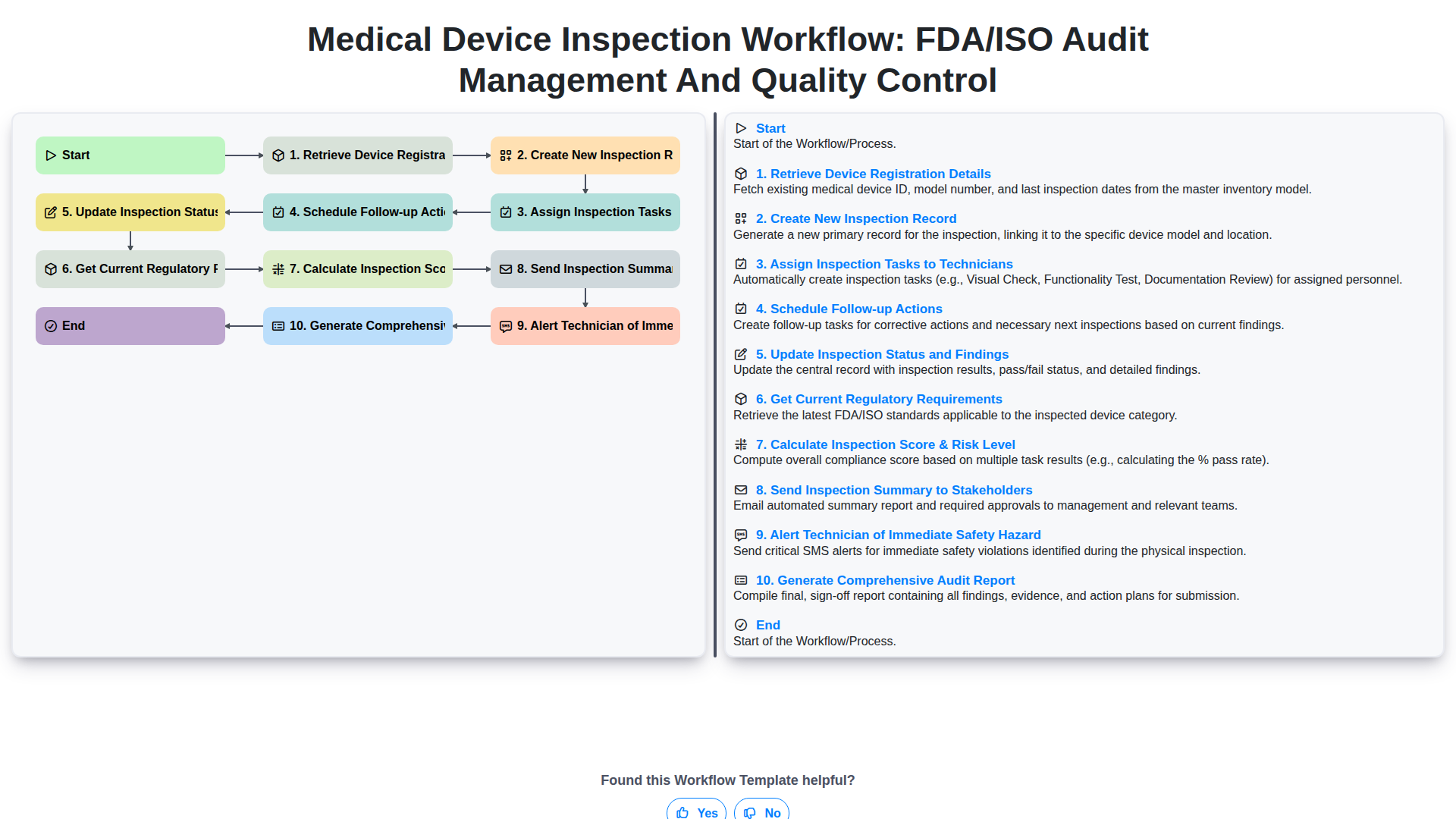
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
Medical Device Inspection Workflow: FDA & Quality Compliance Tracking
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.