Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
Manage your medical device inspections with our comprehensive workflow. From pre-inspection checklists to final report generation, our solution ensures end-to-end audit readiness, maintaining strict FDA and ISO compliance while boosting operational efficiency.
This Template was installed 5 times.
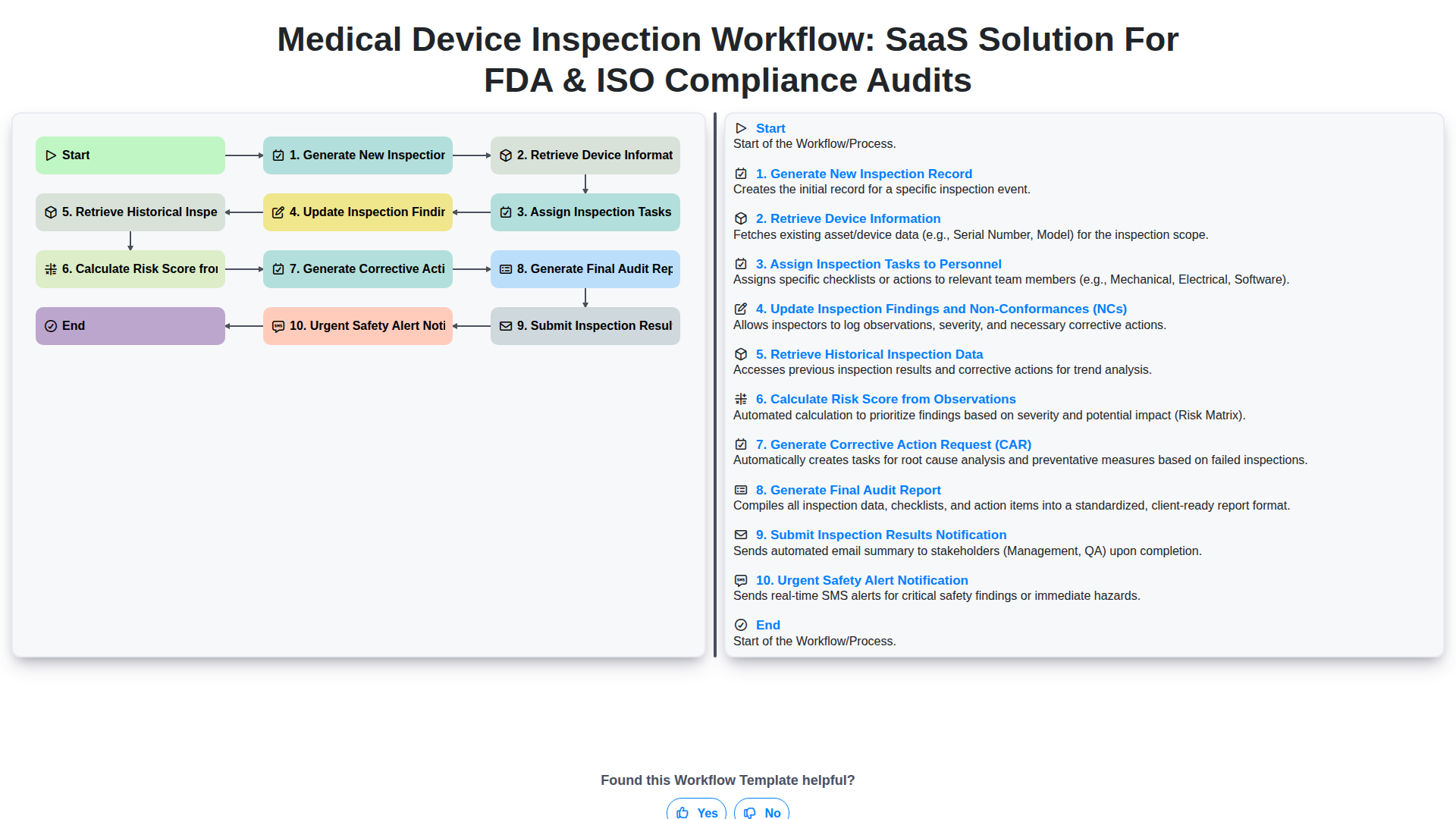
Start
Start of the Workflow/Process.
1. New Inspection Request Initiation
Automatically triggers the inspection process upon request (e.g., scheduled maintenance, ad-hoc check).
2. Inspectable Item Checklist Capture
Allows inspectors to record findings against predefined equipment/process checklists.
3. Corrective Action Implementation & Verification
Used by QA to update status, assign responsibility, and verify closure of identified non-conformances.
4. Historical Inspection Data Retrieval
Pulls past records, failure rates, and prior audit findings for benchmarking.
5. Required Next Step Assignment
Generates follow-up tasks automatically based on inspection findings (e.g., 'Recalibrate X Component').
6. Overall Inspection Risk Score Calculation
Calculates a composite risk score based on the severity and number of failed items found during the inspection.
7. Daily Inspection Summary Report Email
Sends summary reports of completed inspections to management for review.
8. Official Inspection Summary Report Generation
Generates a final, immutable PDF report including scope, checklist results, and corrective actions.
9. Total Findings Count Aggregation
Sums the total number of non-compliant or required entries across the inspection session.
10. Immediate Inspection Completion Notification
Sends an instant SMS alert to supervisor upon successful, critical inspection sign-off.
End
End of the Workflow/Process.
Start of the Workflow/Process.
Automatically triggers the inspection process upon request (e.g., scheduled maintenance, ad-hoc check).
Allows inspectors to record findings against predefined equipment/process checklists.
Used by QA to update status, assign responsibility, and verify closure of identified non-conformances.
Pulls past records, failure rates, and prior audit findings for benchmarking.
Generates follow-up tasks automatically based on inspection findings (e.g., 'Recalibrate X Component').
Calculates a composite risk score based on the severity and number of failed items found during the inspection.
Sends summary reports of completed inspections to management for review.
Generates a final, immutable PDF report including scope, checklist results, and corrective actions.
Sums the total number of non-compliant or required entries across the inspection session.
Sends an instant SMS alert to supervisor upon successful, critical inspection sign-off.
End of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
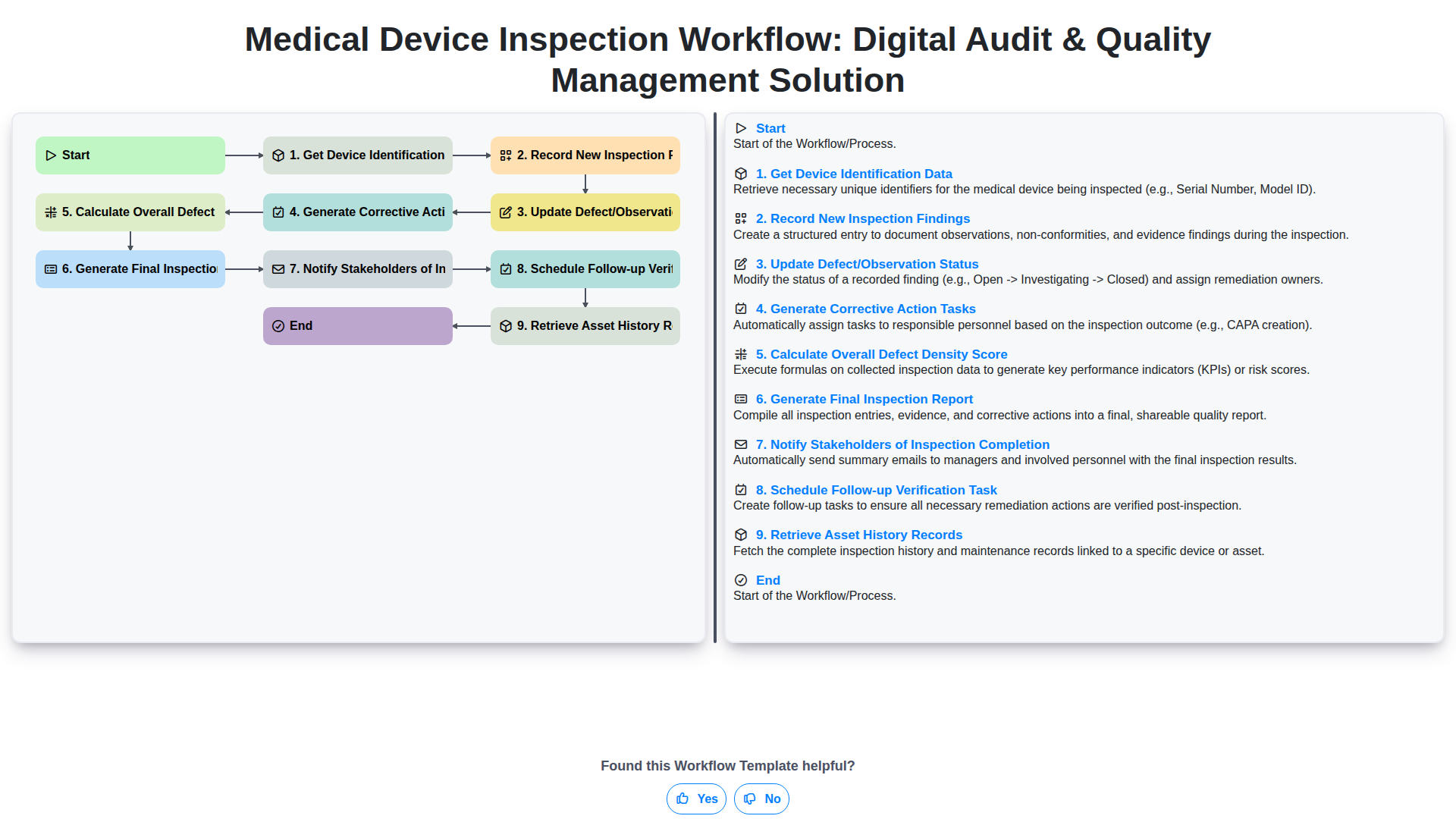
Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
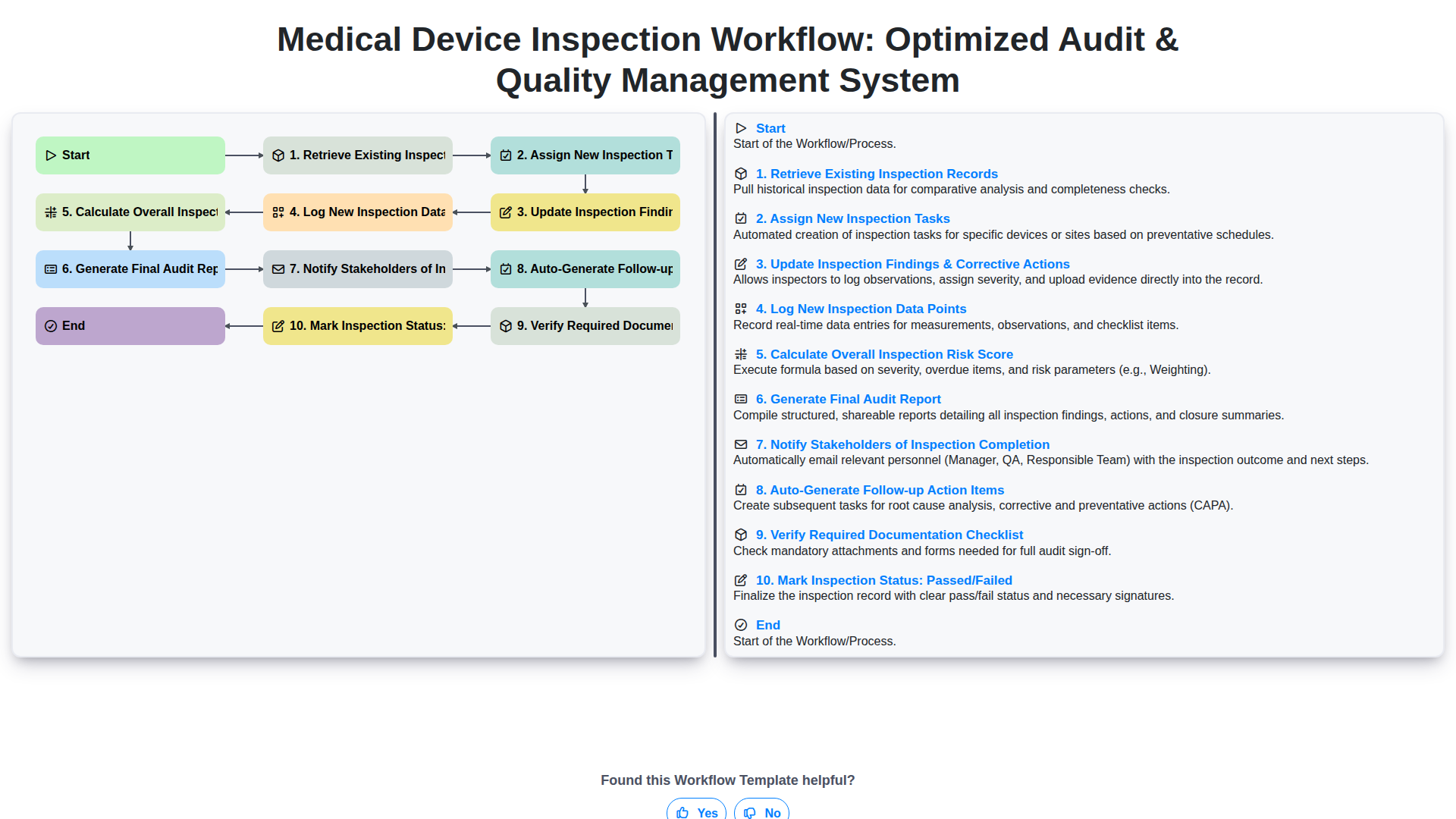
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
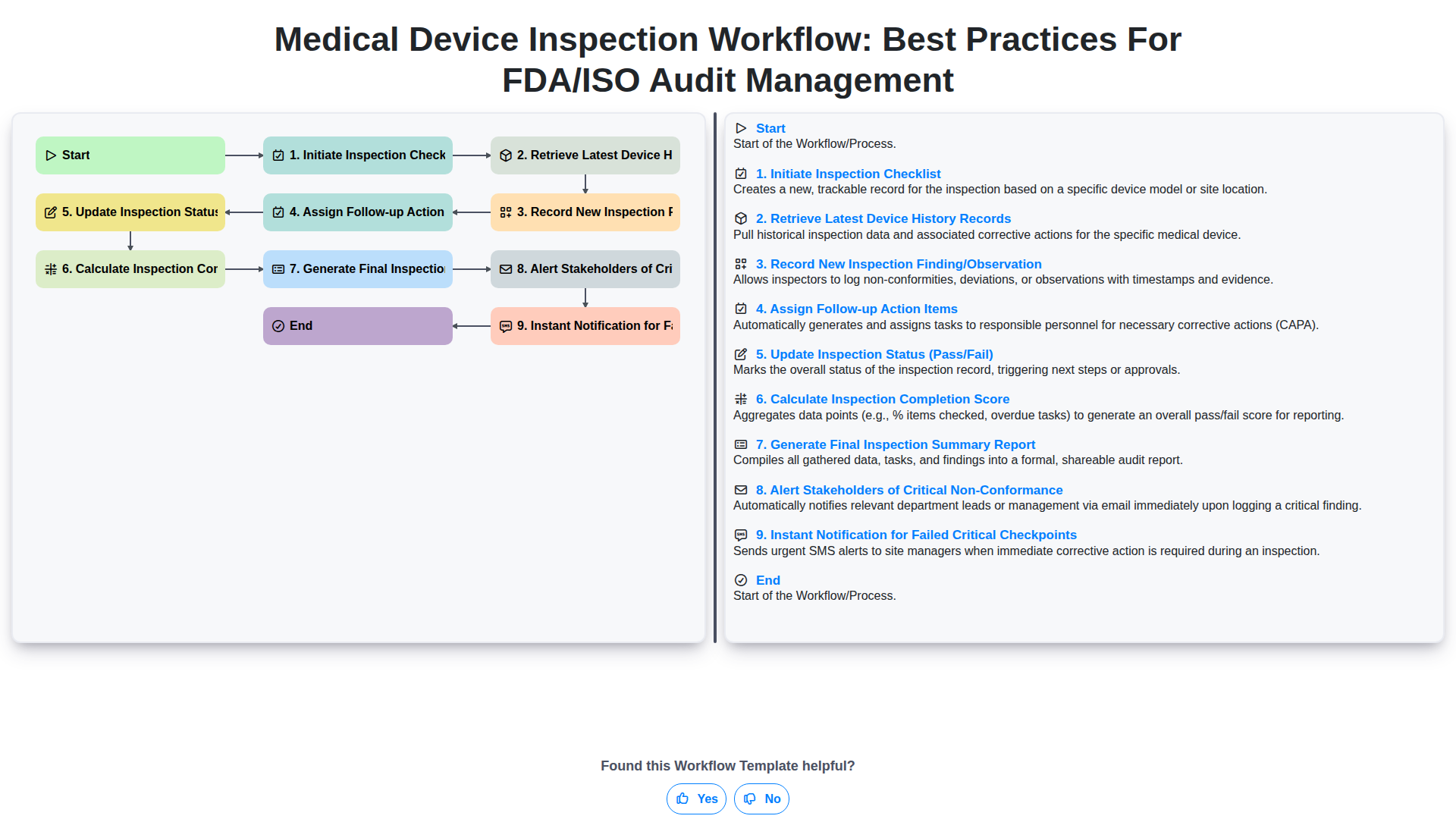
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
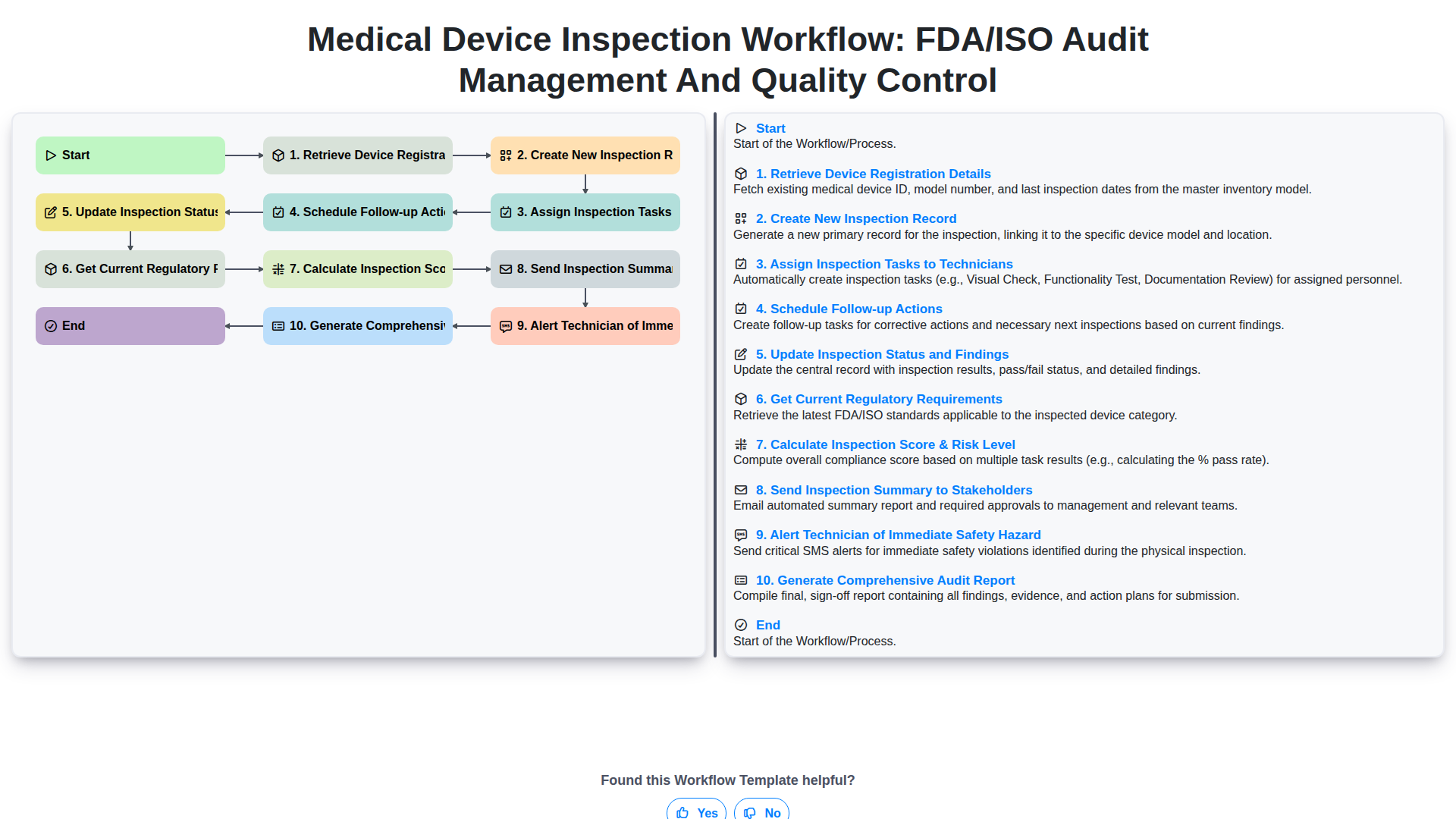
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Streamlined Audit & Compliance Management

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
Medical Device Inspection Workflow: FDA & Quality Compliance Tracking
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.