Pharmaceutical Facility Inspection Workflow: FDA Compliance & Quality Assurance Management
Streamline pharmaceutical inspections with our comprehensive workflow. Ensure continuous FDA compliance and robust quality assurance documentation for your manufacturing facilities. Automate audits, manage deviations, and maintain pristine regulatory records easily.
This Template was installed 4 times.
Start
Start of the Workflow/Process.
1. Initiate Inspection Checklist
Creates a new inspection record for a specific system or area.
2. Retrieve Facility Details
Fetch necessary facility and equipment details from the master data model.
3. Check Previous Inspection Results
Pull historical data to identify areas requiring heightened scrutiny.
4. Assign Inspection Tasks to Team Members
Automatically assign specific inspection checklists to relevant personnel.
5. Update Inspection Status
Mark the current inspection step as complete, recording findings.
6. Log Non-Conformance Findings
Create a new entry detailing any deviations found during the inspection.
7. Schedule Follow-up Actions
Generate and assign remediation tasks for identified non-conformances.
8. Calculate Overall Compliance Score
Automatically score the inspection based on passing/failing criteria.
9. Generate Final Inspection Report
Compiles all data, findings, and corrective actions into a comprehensive report.
10. Send Daily Inspection Summary
Distributes daily summary reports to Quality Assurance and Management.
11. Retrieve Equipment Specifications
Get mandatory operational parameters for the inspected equipment.
End
End of the Workflow/Process.
Start of the Workflow/Process.
Creates a new inspection record for a specific system or area.
Fetch necessary facility and equipment details from the master data model.
Pull historical data to identify areas requiring heightened scrutiny.
Automatically assign specific inspection checklists to relevant personnel.
Mark the current inspection step as complete, recording findings.
Create a new entry detailing any deviations found during the inspection.
Generate and assign remediation tasks for identified non-conformances.
Automatically score the inspection based on passing/failing criteria.
Compiles all data, findings, and corrective actions into a comprehensive report.
Distributes daily summary reports to Quality Assurance and Management.
Get mandatory operational parameters for the inspected equipment.
End of the Workflow/Process.
Found this Workflow Template helpful?
Audit/Inspection Management Solution Demo
Ensure compliance & improve performance! ChecklistGuro streamlines audit/inspection creation, execution, and reporting. Reduce risk, enhance quality, & maintain consistency. Manage it all with our Work OS.
Related Workflow Templates
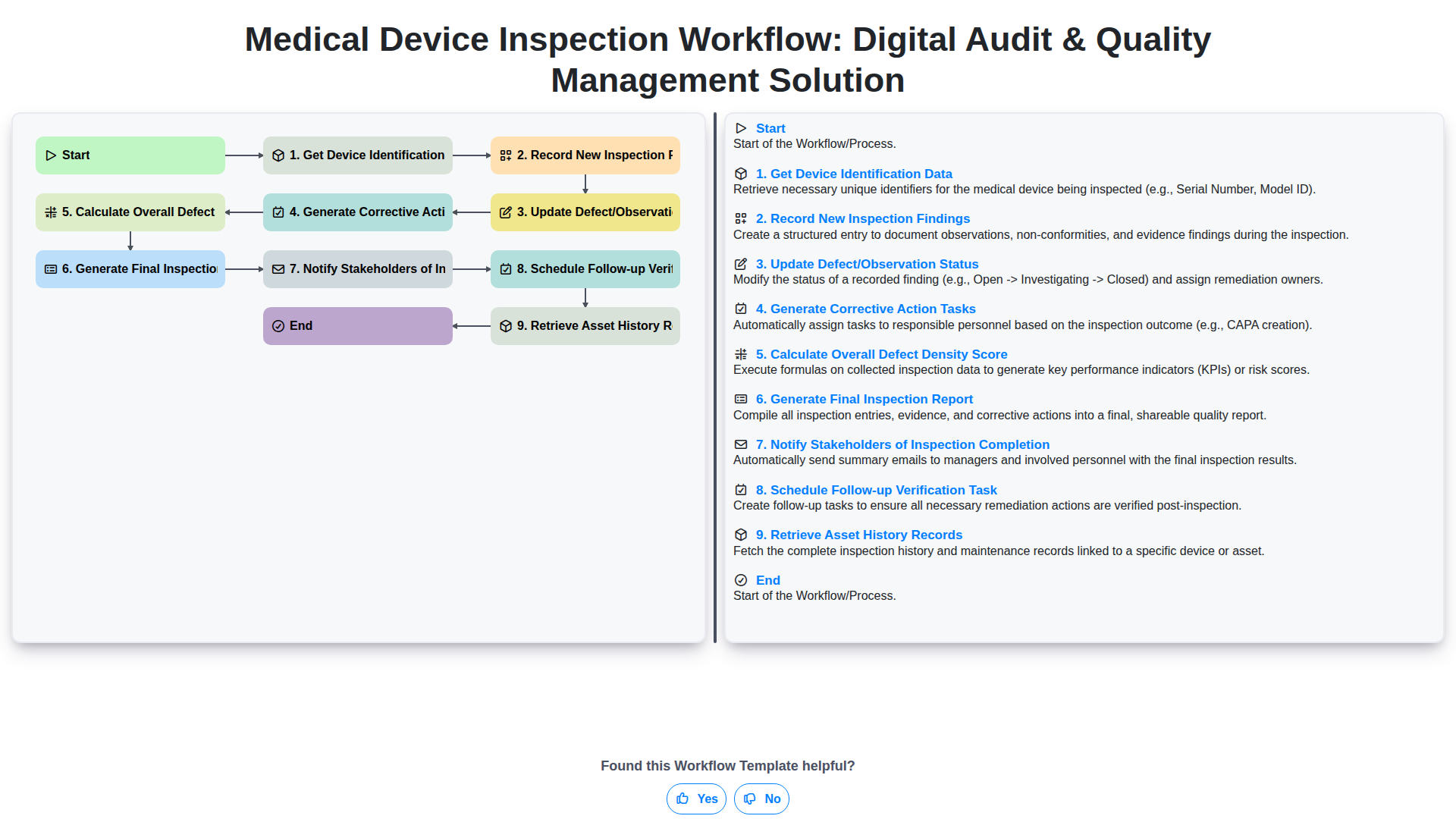
Medical Device Inspection Workflow: Digital Audit & Quality Management Solution
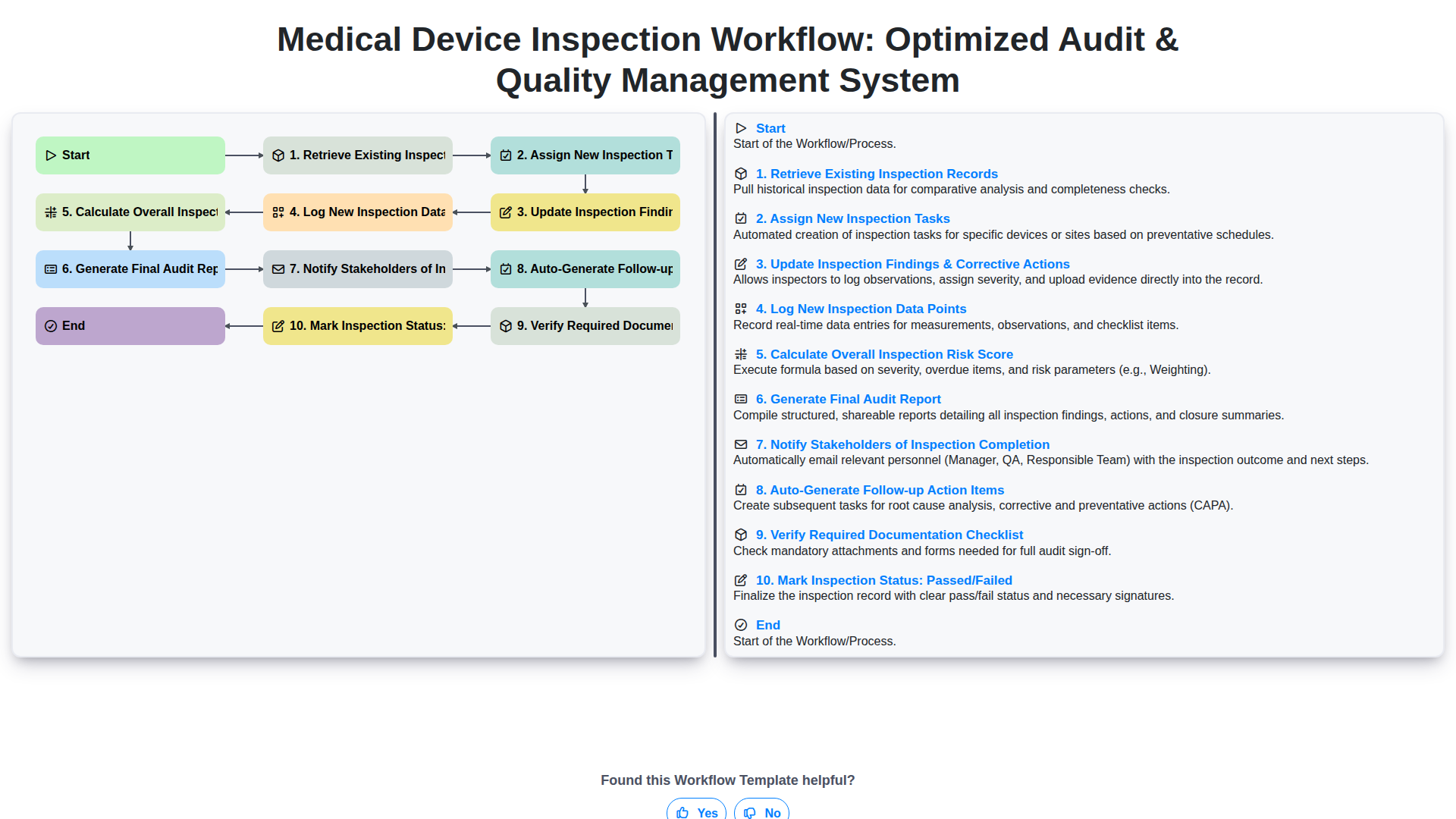
Medical Device Inspection Workflow: Optimized Audit & Quality Management System
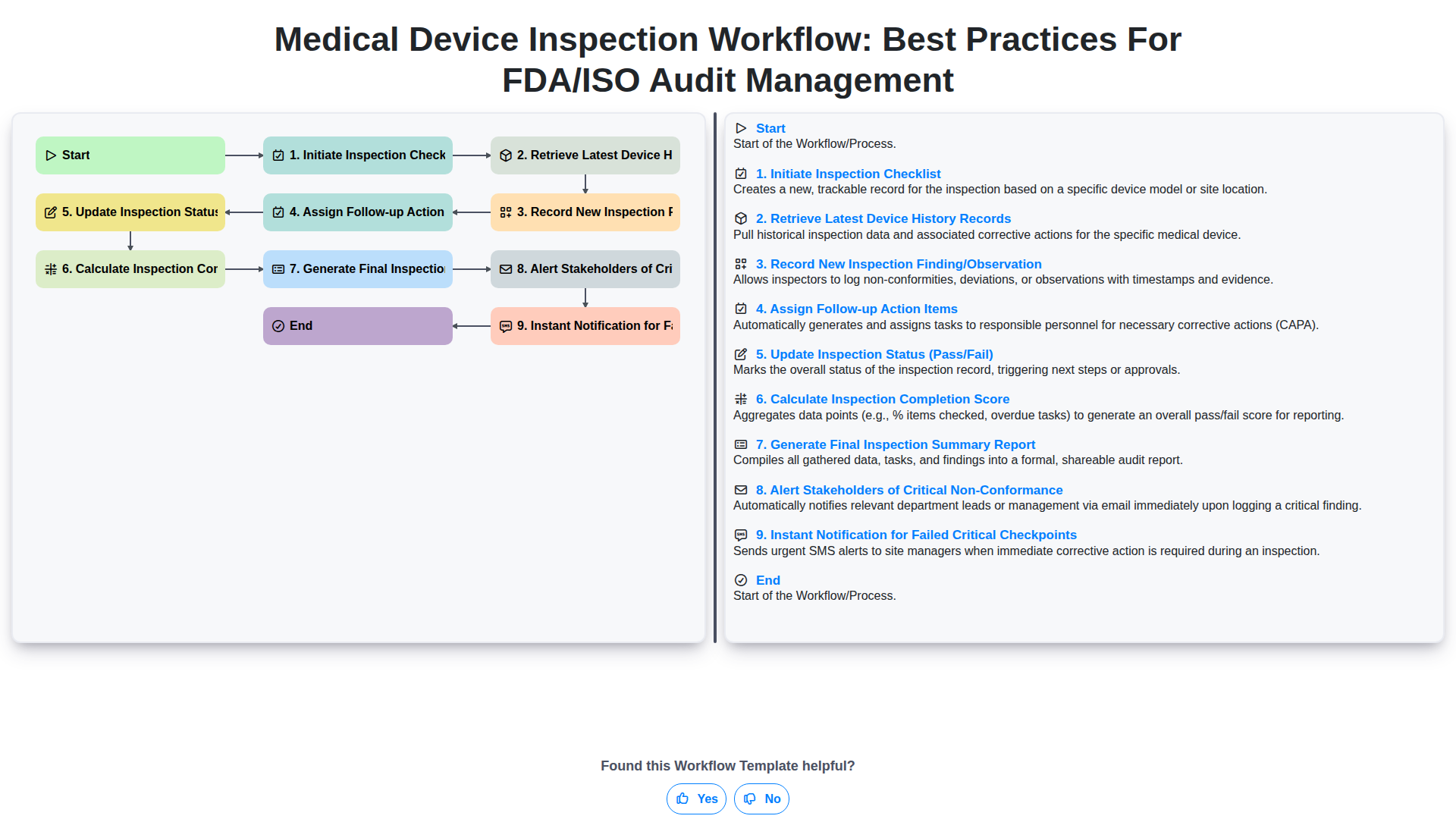
Medical Device Inspection Workflow: Best Practices for FDA/ISO Audit Management
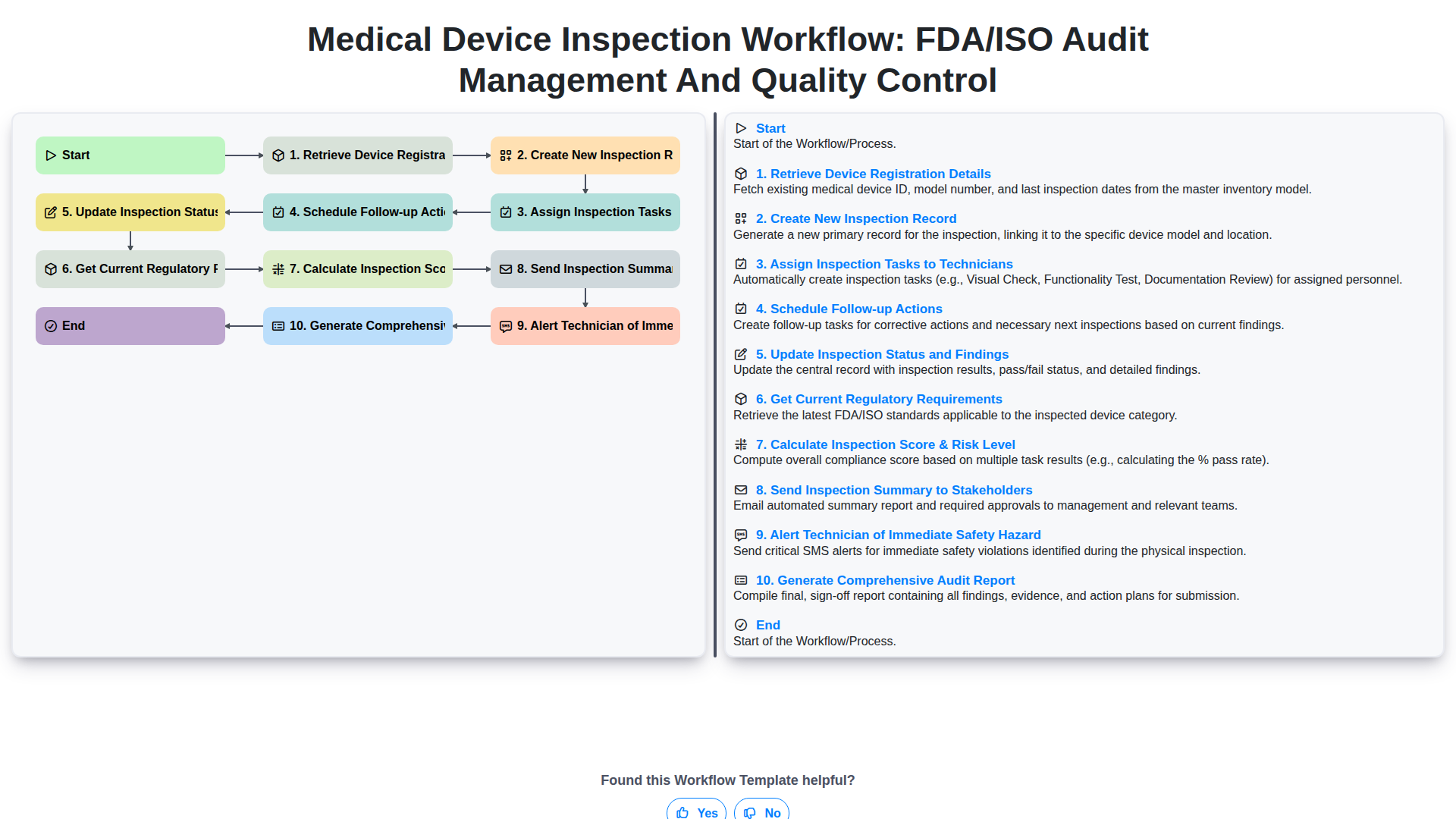
Medical Device Inspection Workflow: FDA/ISO Audit Management and Quality Control
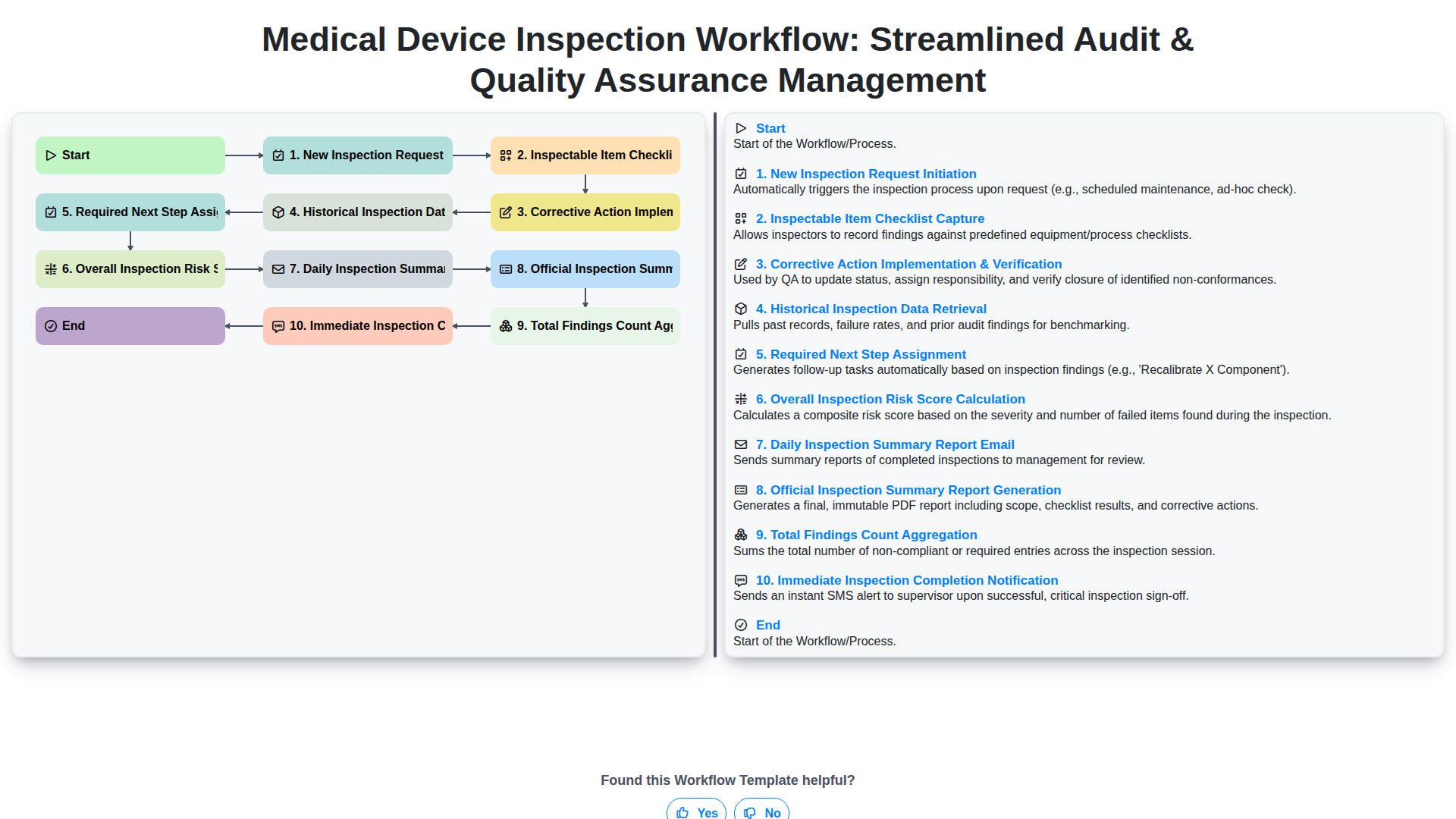
Medical Device Inspection Workflow: Streamlined Audit & Quality Assurance Management
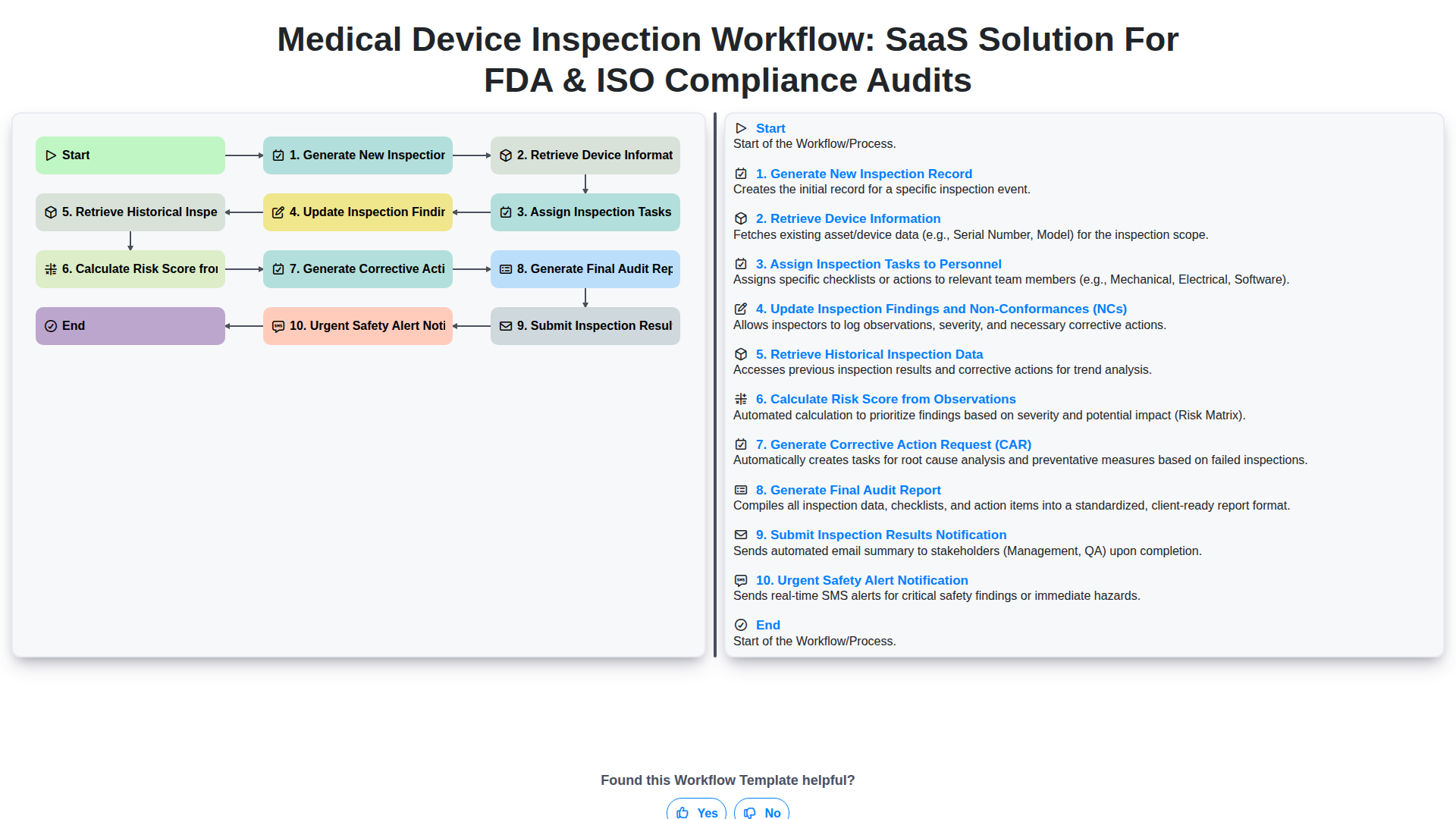
Medical Device Inspection Workflow: SaaS Solution for FDA & ISO Compliance Audits

Medical Device Inspection Workflow: Streamlined Audit & Compliance Management

Medical Device Inspection Workflow: Audit Readiness & Quality Management System
We can do it Together
Need help with
Inspection Management?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.