Manufacturing Process Validation Workflow: A Step-by-Step Guide
Published: 03/30/2026 Updated: 03/31/2026
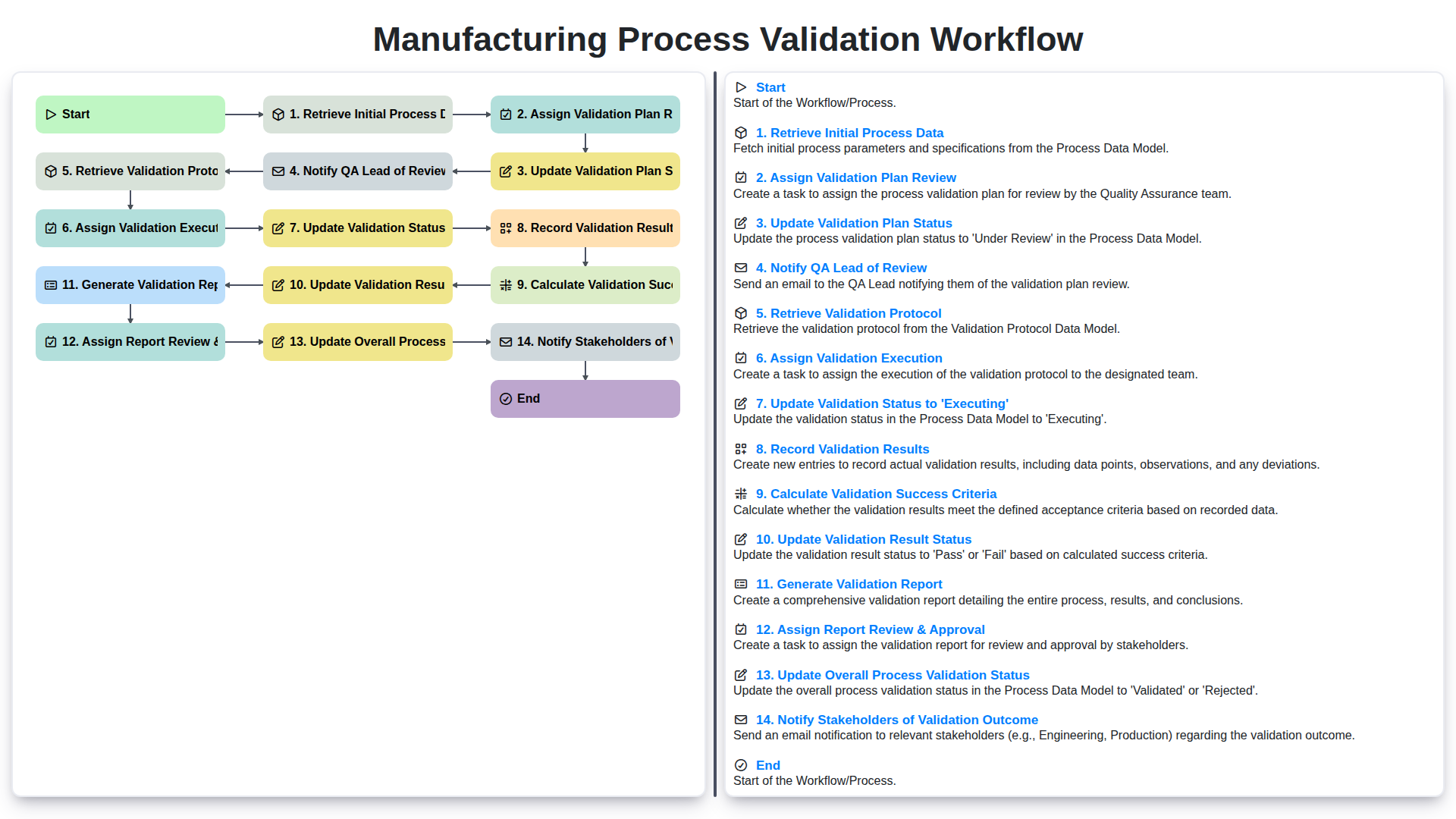
Table of Contents
- Introduction to Manufacturing Process Validation (MPV)
- Why a Structured Workflow is Crucial
- Step 1: Retrieve Initial Process Data
- Step 2: Assign Validation Plan Review
- Step 3: Update Validation Plan Status
- Step 4: Notify QA Lead of Review
- Step 5: Retrieve Validation Protocol
- Step 6: Assign Validation Execution
- Step 7: Validation Status - Moving to 'Executing'
- Step 8: Diligent Record Validation Results
- Step 9: Defining and Calculating Validation Success Criteria
- Step 10: Updating Validation Result Status
- Step 11: Generating the Validation Report
- Step 12: Report Review & Approval - Ensuring Accuracy
- Step 13: Updating Overall Process Validation Status
- Step 14: Notifying Stakeholders of Validation Outcome
- Resources & Links
TLDR: Ensure your manufacturing processes are compliant and reliable with this step-by-step workflow! It guides you from gathering initial data and planning, through execution and reporting, to final approval and stakeholder notification, guaranteeing consistent and documented validation every time.
Introduction to Manufacturing Process Validation (MPV)
Manufacturing Process Validation (MPV) is a critical cornerstone of quality assurance in regulated industries like pharmaceuticals, medical devices, and food production. It's more than just a checklist; it's a systematic, documented process demonstrating that a manufacturing process consistently produces a product meeting pre-determined quality attributes. MPV ensures that your process operates within established parameters, minimizing variability and maximizing the likelihood of producing safe, effective, and reliable products every time. Failing to adequately validate processes can lead to product recalls, regulatory scrutiny, and ultimately, damage to your company's reputation. This blog post will delve into the workflow involved, but first, it's important to understand why MPV is so vital and how it contributes to a robust quality system. It's a proactive approach to quality - identifying and mitigating risks before they impact the final product.
Why a Structured Workflow is Crucial
In manufacturing, validation isn't just about ticking boxes; it's about ensuring consistent product quality and safety. A haphazard, ad-hoc approach to process validation can lead to inconsistencies, delays, and potentially, compromised product. That's where a structured workflow becomes absolutely crucial.
Without a clear, defined workflow, documentation can be incomplete or scattered, making audits a nightmare. Responsibilities become blurred, increasing the risk of errors and missed steps. Furthermore, tracking progress and identifying bottlenecks becomes incredibly difficult, impacting timelines and potentially delaying product launches.
A well-defined workflow, like the Manufacturing Process Validation Workflow we're outlining, provides a roadmap, ensuring each step is completed correctly and consistently. It establishes clear ownership, provides a transparent audit trail, and facilitates efficient communication between teams - ultimately bolstering confidence in your manufacturing process and the products it produces. It's an investment that pays off in reduced risk, improved efficiency, and enhanced product quality.
Step 1: Retrieve Initial Process Data
Before any validation plan can be created, a thorough understanding of the manufacturing process itself is essential. This begins with retrieving the initial process data. This data serves as the baseline for establishing validation parameters and defining acceptance criteria. Key data points to gather include, but are not limited to: process flow diagrams, equipment specifications and maintenance records, raw material specifications, in-process controls, critical process parameters (CPPs), and existing process documentation. This information provides context and helps identify potential risks and areas requiring specific attention during the validation process. This stage is vital for ensuring the validation plan is aligned with the actual manufacturing reality and sets the foundation for a robust and defensible validation.
Step 2: Assign Validation Plan Review
Once the initial process data is gathered and documented, the next crucial step is to assign the Validation Plan Review. This isn't a task for just anyone; it requires a qualified individual or team with a deep understanding of the process, applicable regulations (like GMP, FDA guidelines, etc.), and validation principles.
This reviewer's role is critical. They meticulously examine the Validation Plan to ensure it's comprehensive, technically sound, and aligns with the intended process. They're looking for gaps in the plan, potential risks that haven't been accounted for, and ensuring the proposed validation approach is appropriate for the complexity and criticality of the process. This review may involve questions and clarifications with the original plan authors. Clear documentation of the reviewer's comments and any revisions made to the plan is essential for traceability and audit readiness. The Validation Plan remains 'Pending Review' until this step is successfully completed.
Step 3: Update Validation Plan Status
Once the Validation Plan has been reviewed and approved, the next crucial step is to update its status within the workflow system. This signifies that the plan is ready for execution and removes it from the review queue. The status change typically moves it from 'Review Pending' to 'Approved' or a similar designation. This action triggers downstream actions within the workflow, such as making the Validation Protocol available and initiating the assignment of execution responsibilities. Accurate and timely updates to the Validation Plan Status are vital for maintaining workflow efficiency and ensuring that validation activities progress according to schedule. The system should log the user who performed the update and the timestamp for audit trail purposes.
Step 4: Notify QA Lead of Review
Once the Validation Plan Review is assigned, timely notification to the QA Lead is crucial. This step ensures they're aware of the plan's progress and can provide guidance or intervention if needed. The notification typically includes the assigned reviewer, the validation plan document name, and the date of assignment. This proactive communication fosters collaboration and helps prevent bottlenecks, ensuring the validation process stays on track and maintains quality oversight. Automated notifications, often integrated into the workflow system, are the preferred method to guarantee consistent and immediate awareness.
Step 5: Retrieve Validation Protocol
Once the validation execution has been assigned, the next critical step is retrieving the Validation Protocol. This document serves as the detailed roadmap for the validation execution itself. It outlines the specific procedures, acceptance criteria, test methods, and equipment required to demonstrate that the manufacturing process consistently produces a product meeting predetermined specifications.
The protocol should be readily accessible from a centralized document management system. It's vital to ensure that the version retrieved is the approved and current version, tied directly to the Validation Plan. Discrepancies between the plan and the protocol are immediately flagged and resolved before any execution proceeds. The person assigned to execution should confirm they understand the protocol's requirements and have the necessary training and resources to perform the validation activities as described. A quick review of the protocol's scope and objectives at this stage prevents misunderstandings and ensures the execution aligns with the overall validation goals.
Step 6: Assign Validation Execution
Once the Validation Protocol is retrieved and reviewed, the next critical step is assigning responsibility for its execution. This isn't just about picking someone; it's about ensuring the right personnel are selected based on their expertise, understanding of the process, and familiarity with validation principles.
The assignment process should clearly define the individual(s) accountable for performing the validation activities outlined in the protocol. This includes specifying:
- The assigned validator(s): Document the names and roles of the individuals responsible for executing each step of the protocol.
- Specific tasks: Outline which aspects of the protocol each assigned individual will perform. This prevents overlap and ensures all tasks are covered.
- Timeline: Establish clear start and completion dates for the validation execution phase.
- Resources: Identify any equipment, materials, or software required for the execution and ensure their availability.
Proper assignment is essential for accountability, efficient execution, and ultimately, a robust validation outcome. The documentation of this assignment is a crucial record within the validation workflow.
Step 7: Validation Status - Moving to 'Executing'
Once the Validation Protocol has been successfully retrieved and assigned, the next critical step is to update the Validation Status to 'Executing'. This signifies that the practical validation activities are now underway. This status change triggers several actions within the workflow, primarily alerting assigned personnel that their responsibilities related to execution have commenced. It also provides a clear visual indicator of progress for all stakeholders following the validation process. This transition is a key checkpoint, confirming readiness for the hands-on validation work and preventing premature reporting or interpretation of results. Accurate and timely updates to this status are vital for maintaining workflow integrity and ensuring accountability.
Step 8: Diligent Record Validation Results
Accurate and comprehensive record-keeping during validation execution is absolutely critical. This isn't just about ticking a box; it's about building a robust audit trail that demonstrates the process's reliability. All observations, deviations, and actions taken during the validation execution must be meticulously documented. This includes raw data, any calculations performed, equipment settings, personnel involved, and any unexpected events. Utilize pre-defined templates and standardized forms to ensure consistency and completeness. Any anomalies or deviations from the protocol must be clearly described, along with the rationale behind any corrective actions taken. Photographs, charts, and graphs can be valuable additions to the record to provide visual context and support data interpretation. Remember, the validation record serves as primary evidence for demonstrating compliance and should withstand scrutiny during audits.
Step 9: Defining and Calculating Validation Success Criteria
Defining clear and measurable success criteria is arguably one of the most critical steps in the entire validation process. Without them, you have no objective way to determine if your manufacturing process is truly performing as intended and consistently producing quality products. These criteria aren't just it looks good - they're quantifiable measures tied directly to your process parameters and product specifications.
The process begins with a thorough review of the Validation Plan. This review should identify Critical Process Parameters (CPPs) and Critical Quality Attributes (CQAs). CPPs are the process inputs or controls that must be within an established range to ensure CQAs are met. For each CPP, we must define acceptable limits - the boundaries within which the process can operate without impacting product quality. These limits should be based on risk assessments, historical data, process understanding, and regulatory requirements.
Calculating the success criteria isn't just about setting arbitrary numbers. Consider the following:
- Statistical Significance: Ensure your sample sizes and measurements are statistically significant to accurately represent the process's performance.
- Process Capability: Your success criteria should demonstrate that the process is capable of consistently meeting requirements. This often involves calculating process capability indices like Cp and Cpk.
- Alignment with Specifications: The acceptance criteria must directly align with product specifications and regulatory guidelines.
- Documentation: All success criteria, the rationale behind them, and the calculations used to derive them must be meticulously documented in the Validation Protocol.
Failure to clearly define and calculate validation success criteria can lead to subjective assessments, re-validation efforts, and ultimately, compromised product quality.
Step 10: Updating Validation Result Status
Once the validation execution is complete and all results have been meticulously recorded, the next crucial step is updating the Validation Result Status. This isn't a simple checkbox; it's a formal acknowledgement that the results are available for review and have been processed.
Within the workflow system, the status is typically updated from Executing to a provisional status like Results Recorded or Awaiting Review. This signifies that the data exists and is ready for analysis. This also triggers automated notifications to relevant personnel, such as the Validation Plan Reviewer and QA Lead, alerting them that the results are accessible and ready for evaluation against the pre-defined acceptance criteria. Maintaining accurate and timely status updates ensures transparency and keeps all stakeholders informed of the validation progress. This step also provides an audit trail documenting the transition from execution to review.
Step 11: Generating the Validation Report
Once validation execution is complete and results are meticulously recorded, the next critical step is generating the Validation Report. This report isn't just a summary; it's a comprehensive document demonstrating the entire validation process, findings, and conclusions.
The report should include, but isn't limited to:
- Executive Summary: A concise overview of the validation project's purpose, scope, and key findings.
- Process Description: A clear and detailed explanation of the manufacturing process being validated.
- Validation Plan & Protocol References: Clearly state which plans and protocols this report supports.
- Methodology: Outline the approach used for validation, referencing specific testing methods and acceptance criteria.
- Raw Data & Analysis: Present the recorded results in a clear and understandable format, including all relevant data tables, graphs, and statistical analysis.
- Deviation Handling: Document any deviations encountered during the validation process and explain the corrective actions taken.
- Conclusion: Explicitly state whether the manufacturing process met the pre-defined acceptance criteria and, therefore, is validated.
- Signatures & Dates: Include signatures and dates from the individuals responsible for performing the validation and reviewing the report.
The report must be written in a traceable and auditable format, following established SOPs and regulatory guidelines. Accuracy and completeness are paramount, as this report serves as definitive proof of validation.
Step 12: Report Review & Approval - Ensuring Accuracy
Once the validation report has been generated, it's time for a crucial review and approval phase. This isn't just a formality; it's a final check to ensure the report accurately reflects the validation activities and results, and that all conclusions are supported by the data.
The assigned reviewers, typically a combination of validation engineers, process owners, and potentially representatives from Quality Assurance, meticulously examine the report for completeness, accuracy, and adherence to established protocols and acceptance criteria. They verify that:
- Data Integrity: The reported data aligns with the recorded validation results.
- Conclusion Justification: Conclusions drawn are logically supported by the evidence presented.
- Protocol Adherence: The validation activities were performed as described in the validation protocol.
- Deviation Handling: Any deviations from the protocol are properly documented and justified.
- Regulatory Compliance: The report meets all relevant regulatory requirements.
Upon successful review and confirmation of accuracy, the report is formally approved, signifying completion of the validation process. Any necessary corrections or clarifications identified during the review are addressed and documented before final approval is granted. This step ensures a robust and defensible validation record.
Step 13: Updating Overall Process Validation Status
Once the Validation Report has been reviewed and approved, the final step is to update the overall Process Validation Status. This signifies that the validation exercise is complete and the process is considered validated. The system should automatically reflect this change, transitioning the validation status to "Validated" or a similar conclusive designation. This centralized status provides a clear and consistent view of the entire process validation lifecycle, accessible to all relevant personnel. Furthermore, this final status change should trigger any necessary actions, such as archiving relevant documentation or initiating the scheduled review cycle. This confirmation of validation completion and the subsequent system update are crucial for maintaining accurate records and ensuring ongoing compliance.
Step 14: Notifying Stakeholders of Validation Outcome
Once the Validation Report has been reviewed and approved, the final and crucial step is to formally notify all relevant stakeholders of the validation outcome. This isn't just a courtesy; it's a vital communication piece that ensures transparency and alignment across the organization.
Who are these stakeholders? They typically include:
- Production Team: They need to know the process is validated and ready for routine manufacturing.
- Engineering Team: They're interested in the validation's impact on process design and potential improvements.
- Quality Assurance (QA) Team: They require confirmation of validation success for ongoing process control.
- Management: Provides oversight and ensures regulatory compliance.
- Regulatory Affairs (if applicable): Needed for record-keeping and potential audits.
The notification should be clear, concise, and documented. It should include a summary of the validation results (pass/fail), a reference to the Validation Report, and any key findings or recommendations. Methods for notification can vary - email, a project management system update, or a formal meeting - but documentation of the notification itself is essential. This demonstrates a controlled and auditable process.
Resources & Links
- U.S. Food and Drug Administration (FDA) : The FDA provides extensive guidance and regulations regarding process validation, particularly for pharmaceutical manufacturing. This is a primary source for understanding regulatory requirements.
- International Society for Pharmaceutical Engineering (ISPE) : ISPE offers comprehensive resources, publications, and training on process validation, including best practices and industry standards. Their 'Process Validation Guidance' is highly regarded.
- American Society for Quality (ASQ) : ASQ offers quality management resources, training, and certifications related to process validation and quality assurance, including standards and methodologies.
- Pharmaceutical Technology : Offers a wide range of articles and insights on pharmaceutical manufacturing, including process validation approaches, technologies, and regulatory updates.
- EMC2 : EMC2 provides process validation services and consulting. Their website contains articles and whitepapers explaining aspects of MPV and related concepts.
- Quality Digest : A website dedicated to quality management and continuous improvement, offering articles and insights relevant to manufacturing process validation.
- National Institute of Standards and Technology (NIST) : NIST provides standards and guidelines related to measurement and validation processes that can be relevant to MPV, although not directly focused on validation itself. It's useful for understanding measurement science.
- GMP Compliance : A resource focused on Good Manufacturing Practices (GMP) and regulatory compliance, offering articles and training materials relating to process validation.
Frequently Asked Questions
What is manufacturing process validation?
Manufacturing process validation is the documented evidence that a specific process will consistently produce a product meeting its predetermined specifications and quality attributes.
What are the three main stages of the validation lifecycle?
The process validation lifecycle typically consists of three stages: Process Design, Process Qualification (including IQ, OQ, and PQ), and Continued Process Verification (CPV).
What does IQ, OQ, and PQ stand for in validation?
IQ stands for Installation Qualification (verifying equipment is installed correctly), OQ stands for Operational Qualification (verifying equipment operates within defined limits), and PQ stands for Performance Qualification (verifying the process performs consistently under production conditions).
Why is a validation protocol important?
A validation protocol is a critical document that outlines the testing parameters, sampling plans, acceptance criteria, and procedures to be followed to ensure the validation is performed scientifically and meets regulatory standards.
How often should process validation be performed?
While initial validation occurs during new process setup, ongoing monitoring through Continued Process Verification (CPV) is required to ensure the process remains in a state of control throughout its entire lifecycle.
What should be done if a validation test fails?
If a validation test fails, a formal investigation (often using Root Cause Analysis) must be conducted to identify the cause of the deviation, implement corrective actions, and potentially repeat the validation steps.
Found this Article helpful?
Manufacturing Management Solution Demo
Stop chasing paperwork and wrestling with disconnected systems! Our solution brings clarity and control to your production process. From standard operating procedures (SOPs) and equipment maintenance to quality control and inventory tracking, we simplify every step.
Related Articles
Kaizen Checklist Template: Your Path to Continuous Improvement

Machine Safety Audit Checklist: Your Guide to a Safer Manufacturing Workplace
Mastering Supplier Performance: Your Free Supplier Scorecard Checklist Template
Your Ultimate RCM Checklist Template: A Step-by-Step Guide

LPA Checklist Template: Your Guide to Layered Process Audits

Your Ultimate Lockout/Tagout (LOTO) Checklist Template

Your Ultimate FSC 22000 Checklist Template
Quality Control Inspection Checklist Template: Prevent Defects & Improve Manufacturing Processes
We can do it Together
Need help with
Checklists?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.