Supplier Quality Control Workflow: A Step-by-Step Guide
Published: 03/30/2026 Updated: 03/31/2026
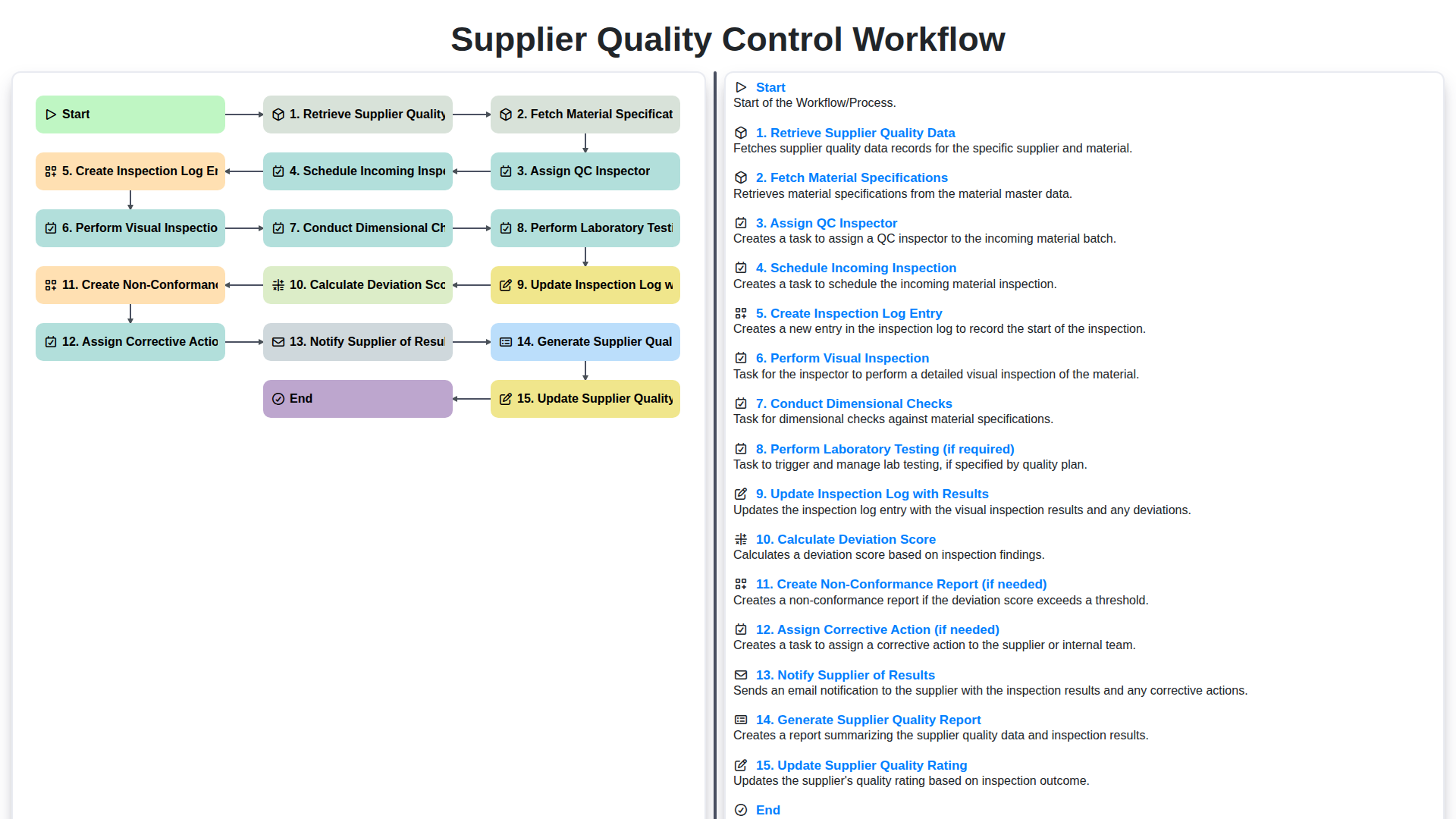
Table of Contents
- Introduction: Why Supplier Quality Control Matters
- 1. Retrieve Supplier Quality Data
- 2. Fetch Material Specifications
- 3. Assign a Qualified QC Inspector
- 4. Schedule the Incoming Inspection
- 5. Create an Inspection Log Entry
- 6. Perform a Thorough Visual Inspection
- 7. Conduct Precise Dimensional Checks
- 8. Laboratory Testing: When and Why
- 9. Document Results: Updating the Inspection Log
- 10. Calculating the Deviation Score
- 11. Identifying Issues: Creating Non-Conformance Reports
- 12. Assigning Corrective Actions
- 13. Supplier Communication: Notifying of Results
- 14. Ongoing Evaluation: Generating Supplier Quality Reports & Updating Ratings
- Resources & Links
TLDR: Ensure consistent, high-quality materials with this workflow! It covers everything from initial data collection and inspection scheduling to lab testing, deviation scoring, non-conformance reporting, corrective actions, supplier notifications, and performance reporting - ultimately optimizing your supplier quality and minimizing risks.
Introduction: Why Supplier Quality Control Matters
The quality of your finished product is only as good as the quality of its components. That's why a robust Supplier Quality Control (SQC) workflow isn't just a "nice-to-have" - it's a vital cornerstone of operational excellence and a key differentiator in today's competitive market. Relying solely on internal quality checks isn't enough; proactively managing the quality of materials and components sourced from your suppliers directly impacts everything from product performance and customer satisfaction to cost savings and brand reputation.
Poor supplier quality can lead to a cascade of issues: costly rework or scrap, production delays, increased warranty claims, and ultimately, damage to your company's credibility. A well-defined SQC workflow shifts the responsibility for quality earlier in the process, minimizing risk and ensuring that only materials meeting your stringent standards enter your production line. This proactive approach protects your bottom line, strengthens your brand, and fosters stronger, more reliable partnerships with your suppliers. Let's explore how a structured workflow can help you achieve this.
1. Retrieve Supplier Quality Data
Gathering reliable supplier quality data is the foundational step in ensuring the integrity of your materials and finished products. This initial phase involves pulling information from multiple sources to build a comprehensive picture of a supplier's performance. We typically draw data from several avenues: past inspection records - both internal and external; supplier audits and questionnaires; certificates of analysis (COAs); and any available quality performance metrics shared directly by the supplier. Accuracy and completeness at this stage are crucial; incomplete or inaccurate data can skew subsequent assessments and lead to flawed decisions. A centralized, accessible database is ideal for storing and managing this information, allowing for easy retrieval and analysis. The data collected should include details such as material type, lot numbers, supplier name, and any relevant historical quality issues.
2. Fetch Material Specifications
Before a single component or raw material can be deemed acceptable, we need to know exactly what it should be. This is where fetching material specifications comes in. This isn't just about having a document; it's about accessing the correct and current specifications.
Here's what's involved:
- Locating Specifications: We maintain a centralized repository (often a PLM or ERP system) where all approved material specifications are stored. The process involves retrieving the specific document linked to the incoming material.
- Verification: The retrieved specification is meticulously checked against the purchase order and material certificate to ensure it corresponds to the expected material. Any discrepancies trigger immediate clarification and potential hold.
- Version Control: Material specifications evolve. We strictly adhere to version control, ensuring the QC inspector uses the latest approved revision. Outdated specifications can lead to incorrect assessments and compromised product quality.
- Accessibility: The specifications must be readily accessible to the assigned QC inspector before the inspection begins. This prevents delays and ensures consistent evaluation.
- Understanding Requirements: The QC inspector needs a thorough understanding of the specifications - not just a cursory glance. Training and readily available resources are essential.
3. Assign a Qualified QC Inspector
Assigning the right Quality Control (QC) Inspector is crucial for ensuring the integrity of your supplier quality control process. This isn't just about filling a role; it's about matching the inspector's skills and expertise to the specific material and potential risks involved.
Consider these factors when assigning an inspector:
- Material Expertise: Does the inspector have experience with the specific material being received? Certain materials (e.g., polymers, composites, specialized alloys) require a deeper understanding of their properties and potential failure modes.
- Relevant Training & Certifications: Ensure the inspector possesses the necessary training and certifications relevant to the inspection requirements (e.g., SPC, GD&T, specific industry standards).
- Availability & Workload: Balance the urgency of the inspection with the inspector's current workload to avoid rushed or compromised assessments.
- Calibration and Equipment Proficiency: The inspector must be comfortable and proficient with the measuring tools and equipment needed for the inspection.
- Documentation Skills: A good QC Inspector should be able to accurately and clearly document inspection findings.
Proper assignment contributes significantly to the overall accuracy and effectiveness of your Supplier Quality Control Workflow.
4. Schedule the Incoming Inspection
Scheduling the incoming inspection is a crucial step to ensure a smooth and efficient receiving process. It's more than just putting it on a calendar; it's about optimizing resources and minimizing delays. Here's what goes into a well-planned schedule:
- Prioritization: Not all incoming shipments are created equal. Consider factors like criticality of the material (impact on production), supplier history (risk assessment), and quantity. High-risk or critical materials should be prioritized.
- Resource Availability: Verify the QC Inspector's availability and workload. Avoid scheduling inspections that will create bottlenecks or extend lead times.
- Material Readiness: Ensure the material is accessible and properly identified before the scheduled inspection time. A clear labeling system is essential.
- Communication: Inform relevant teams (receiving, production planning) about the inspection schedule to avoid disruptions.
- Lead Time Considerations: Factor in the time required for the inspection itself - visual inspection, dimensional checks, and potential laboratory testing.
- Inspection Bay Allocation: If you have dedicated inspection bays, reserve one for the scheduled inspection.
5. Create an Inspection Log Entry
Detailed and accurate record-keeping is paramount in any successful quality control process. Creating an inspection log entry is a crucial step in the Supplier Quality Control Workflow, serving as a single source of truth for all inspection activities related to a specific supplier and material.
This entry should contain key information including:
- Supplier Name & ID: Clearly identifies the supplier being assessed.
- Material Name & Part Number: Specifies the material being inspected.
- Lot/Batch Number: Allows traceability back to the supplier's production run.
- Date & Time of Inspection: Provides a timestamp for the inspection.
- QC Inspector: Records who performed the inspection.
- Inspection Type: Indicates the type of inspection (e.g., Incoming Inspection, First Article Inspection).
- Initial Observations: A brief note of the inspector's initial assessment upon receiving the material.
- Reference Documents: Links or references to relevant material specifications, drawings, and other pertinent documents.
This log entry acts as a central repository of information, facilitating analysis, trending, and ultimately, continuous improvement of supplier quality. It's also vital for audit trails and demonstrating compliance with quality standards.
6. Perform a Thorough Visual Inspection
A crucial first step in assessing incoming materials is a meticulous visual inspection. This isn't just a quick glance; it's a detailed examination of each component or batch against pre-defined acceptance criteria. Look for any obvious defects such as scratches, dents, discoloration, rust, burrs, or other surface imperfections. Assess the overall cleanliness and packaging integrity. Consistent visual cues can often indicate larger systemic issues with the supplier's process. This step helps identify immediately rejectable items, preventing them from progressing further into the production cycle and potentially causing significant downstream problems. Document any observations made during this inspection in the inspection log, including photos where applicable.
7. Conduct Precise Dimensional Checks
Dimensional checks are critical to ensure the received materials meet the specified engineering requirements. This stage involves meticulously measuring key dimensions of the incoming materials using calibrated instruments such as calipers, micrometers, gauges, and coordinate measuring machines (CMMs). We don't just take one measurement; multiple readings are taken at various points on the part to account for potential variations. These measurements are then compared to the material specifications retrieved earlier in the workflow. Any discrepancies, even minor ones, are carefully documented in the inspection log. The level of precision required during dimensional checks is determined by the criticality of the dimension and its impact on the final product's performance and safety. The inspection log will record the instrument used, the measurement values, and a clear indication of whether the part is within tolerance.
8. Laboratory Testing: When and Why
Laboratory testing isn't always necessary, but it's a crucial safety net within our Supplier Quality Control workflow. It's triggered when visual inspection and dimensional checks raise concerns, or when material specifications explicitly require it. This might include situations where we suspect material composition isn't as declared, or when performance characteristics require more rigorous assessment.
The types of lab testing vary significantly depending on the material and its intended use. Examples include:
- Chemical Analysis: Verifying material composition and identifying potential contaminants.
- Mechanical Testing: Assessing strength, hardness, ductility, and other mechanical properties.
- Physical Property Testing: Evaluating characteristics like density, melting point, and electrical conductivity.
- Microscopic Analysis: Examining material microstructure to identify defects or inconsistencies.
We utilize accredited, third-party laboratories for objective and reliable results. The decision to implement laboratory testing is never taken lightly, as it adds time and cost to the process. However, the assurance of quality and compliance it provides is essential for maintaining product integrity and mitigating potential risks.
9. Document Results: Updating the Inspection Log
Accurate and thorough documentation is the cornerstone of any successful quality control process. After the physical inspection and any required laboratory testing are complete, meticulously updating the inspection log is paramount. This isn't just about recording that something happened, but what happened, and with what level of detail.
Each entry should include the date and time of the inspection, the specific lot number and material being assessed, the inspector's name and signature (or electronic equivalent), and a clear, concise description of the results. For visual inspections, detail any deviations from expected appearance. Dimensional checks require precise measurements and tolerances. Laboratory test results should be attached or referenced directly.
Use consistent terminology and standardized formats within the log to ensure clarity and facilitate analysis later on. Photographs and other visual aids are invaluable in documenting defects or anomalies. Finally, remember to clearly indicate any observations that fall outside the defined acceptance criteria - these are the flags that trigger further investigation and potential non-conformance reporting. A well-maintained inspection log serves as a comprehensive record for traceability and continuous improvement.
10. Calculating the Deviation Score
The Deviation Score is a crucial metric for quantifying a supplier's performance and identifying areas needing improvement. It's a numerical representation of how closely the supplier's delivered materials adhere to specified requirements. This isn't a simple pass/fail; it's about understanding the degree of deviation.
The calculation itself involves weighting different aspects of the inspection. For example, a dimensional deviation critical to functionality might carry a higher weight than a minor cosmetic blemish. We typically use a point-based system.
Here's a simplified breakdown of how it works:
- Define Critical Parameters: Identify the key material characteristics (dimensions, chemical composition, mechanical properties, etc.) that have the greatest impact on product quality and performance.
- Establish Tolerance Limits: For each parameter, define acceptable limits of deviation from the specified value (e.g., ±0.1mm for a dimension, +/- 0.5% for a chemical composition).
- Assign Weights: Assign a numerical weight to each parameter based on its criticality. Critical parameters receive higher weights.
- Calculate Deviation Points: For each inspection finding, calculate the deviation from the specification. The magnitude of the deviation is converted into points. A small deviation gets few points; a large deviation gets many. The specific conversion might be linear (e.g., 1 point per 0.01mm deviation) or use a more complex formula to account for non-linear risk.
- Sum Weighted Points: Multiply the points for each deviation by its assigned weight and sum the results to obtain the total Deviation Score.
Example:
| Parameter | Specification | Actual Value | Deviation | Weight | Points | Weighted Points |
|---|---|---|---|---|---|---|
| Diameter | 10.00 mm ± 0.05 mm | 10.08 mm | 0.08 mm | 5 | 4 | 20 |
| Hardness | 55 HRC ± 3 HRC | 56 HRC | 1 HRC | 3 | 2 | 6 |
| Surface Finish | Ra 1.6 µm | Ra 1.8 µm | 0.2 µm | 1 | 1 | 1 |
Total Deviation Score: 27
A higher Deviation Score indicates a larger degree of non-conformance. The threshold for triggering a Non-Conformance Report is pre-defined and based on company policies and risk tolerance. This scoring system provides a quantifiable and objective way to assess supplier quality performance and drive continuous improvement.
11. Identifying Issues: Creating Non-Conformance Reports
When the inspection process uncovers deviations from established specifications, a Non-Conformance Report (NCR) is essential. This isn't just about documenting a problem; it's about initiating a structured response. The NCR meticulously details the nature of the non-conformance: what was found, where it was found (lot number, material ID, etc.), and the severity of the deviation based on the calculated Deviation Score.
A clear and comprehensive NCR ensures everyone involved understands the issue and its impact. It serves as a formal record of the problem, providing a traceable audit trail. The report should include photos or other visual evidence, and link directly to the inspection log entry and the relevant material specifications. This detailed documentation is crucial for root cause analysis and preventing future occurrences. The NCR isn't a blame game; it's a tool for improvement and a commitment to quality.
12. Assigning Corrective Actions
Once a non-conformance report is created, the focus shifts to rectifying the identified issues. This stage involves assigning corrective actions to the responsible parties, which can include individuals within your organization (e.g., engineering, procurement, production) or, in some cases, require collaboration with the supplier.
Clearly define the corrective action plan, outlining specific tasks, timelines, and accountable individuals. Document these details meticulously within the non-conformance report or a related tracking system. Examples of corrective actions could include process adjustments, retraining personnel, modifying supplier requirements, or implementing stricter quality controls at the supplier's facility.
The assigned individuals are then responsible for implementing and tracking the corrective action's progress, ensuring it addresses the root cause of the non-conformance and prevents recurrence. Regular follow-up and verification are crucial to validate the effectiveness of the corrective actions and confirm that the issue has been resolved.
13. Supplier Communication: Notifying of Results
Once the inspection process is complete and all data is compiled, timely and transparent communication with the supplier is crucial. This isn't just about informing them of the outcome; it's about fostering a collaborative relationship and encouraging continuous improvement.
Our workflow dictates that we notify the supplier of the inspection results promptly, typically within [Specify Timeframe, e.g., 24-48 hours]. This notification includes a summary of findings, highlighting any deviations from specifications and the calculated deviation score. We prioritize clear and concise language, avoiding technical jargon whenever possible.
The method of notification varies depending on the supplier's preference and established communication protocols. This can include email, a dedicated supplier portal, or even a phone call for more complex situations. Regardless of the method, documentation of the notification (date, time, recipient, and summary of information shared) is recorded in the inspection log.
Furthermore, we offer opportunities for dialogue. Suppliers are encouraged to ask questions and provide explanations regarding any discrepancies. This interactive approach allows for a better understanding of the issues and can lead to collaborative solutions to prevent future occurrences. We view this as a partnership - their success is tied to our quality, and our communication is vital to achieving that shared goal.
14. Ongoing Evaluation: Generating Supplier Quality Reports & Updating Ratings
The journey towards consistent, high-quality materials doesn't end with a single inspection. Continuous monitoring and evaluation of supplier performance are crucial for long-term success. This is where robust supplier quality reporting and rating systems come into play.
Generating Supplier Quality Reports: Regular reports, compiled from inspection log entries and deviation scores, provide a comprehensive overview of each supplier's adherence to specifications. These reports should include key performance indicators (KPIs) such as:
- Non-Conformance Rate: Percentage of inspections resulting in non-conformances.
- Deviation Score Trends: How the deviation score has changed over time, highlighting areas of improvement or decline.
- Corrective Action Effectiveness: How successfully supplier-implemented corrective actions addressed previously identified issues.
- Inspection Cycle Time: The average time taken to complete an inspection, revealing potential bottlenecks.
These reports aren't just for internal use; sharing summarized versions with suppliers (appropriately redacted for confidentiality) fosters transparency and encourages collaboration.
Updating Supplier Quality Ratings: The data from these reports directly feeds into your supplier quality rating system. A tiered rating system (e.g., Gold, Silver, Bronze) provides a clear, quantifiable measure of supplier performance. Consider these factors when assigning ratings:
- Deviation Score: A lower deviation score generally indicates a higher rating.
- History of Non-Conformances: Repeat offenders should be penalized.
- Responsiveness to Issues: How quickly and effectively the supplier addresses concerns and implements corrective actions.
- Overall Performance Trend: An upward trend reflects positive progress and contributes to a higher rating.
Regularly updating these ratings - ideally quarterly or annually - ensures the system accurately reflects current performance and provides a foundation for strategic supplier management decisions, including potential audits, increased scrutiny, or even the exploration of alternative sourcing options.
Resources & Links
- ISO (International Organization for Standardization) : Provides standards related to quality management, including ISO 9001 which is foundational for supplier quality management systems. Explore relevant standards and guidelines.
- ASQ (American Society for Quality) : A leading professional organization offering resources, training, and certifications related to quality control and improvement, including supplier quality.
- NIST (National Institute of Standards and Technology) : Provides metrology standards and guidance for accurate measurements, vital for dimensional checks and laboratory testing within the workflow.
- ASQ Six Sigma : Provides resources and training related to Six Sigma methodologies which can be applied to improve supplier quality control processes and reduce variability.
- Quality America : Offers quality inspection services and resources, illustrating best practices in visual inspection and dimensional checks that are part of the workflow.
- Association for Professionals in Quality : Offers resources, training, and certifications for quality professionals, including those involved in supplier quality management.
- Reliability Solutions : Focuses on reliability engineering and quality assurance, with insights applicable to ensuring supplier quality and product durability.
- AIAG (Automotive Industry Action Group) : Develops quality standards and best practices primarily for the automotive industry, which are frequently adopted in other sectors. Their CQI-9 'Receiving Inspection' is particularly relevant.
- SAE International : Provides standards and resources for the engineering and mobility industries, including quality and reliability aspects relevant to supplier management.
- BSCI (Business Social Compliance Initiative) : While focused on social compliance, their processes around supplier audits and documentation can offer valuable insights into structured supplier evaluation, a key component of quality control.
Frequently Asked Questions
What is a Supplier Quality Control (SQC) workflow?
A Supplier Quality Control workflow is a structured, step-by-step process used by companies to evaluate, monitor, and manage the quality of goods and services provided by their vendors to ensure they meet specific standards and requirements.
What are the key stages of a typical supplier quality control process?
The key stages generally include supplier selection and qualification, incoming material inspection, in-process monitoring, final product inspection, and continuous performance evaluation/auditing.
Why is supplier auditing important in the workflow?
Auditing is crucial to verify that a supplier's internal processes, facilities, and management systems are capable of consistently producing high-quality products and complying with regulatory or industry standards.
How can companies handle non-conforming goods found during inspection?
When non-conforming goods are identified, companies should initiate a Corrective Action Plan (CAPA), which involves documenting the defect, notifying the supplier, determining the root cause, and implementing measures to prevent recurrence.
What metrics should be used to track supplier performance?
Key performance indicators (KPIs) often include defect rates, On-Time Delivery (OTD) percentage, frequency of corrective actions, audit scores, and responsiveness to quality issues.
How does a robust SQC workflow reduce overall operational costs?
A robust workflow reduces costs by minimizing product recalls, decreasing waste from defective materials, preventing production downtime, and reducing the administrative burden of managing frequent quality disputes.
Found this Article helpful?
Manufacturing Management Solution Demo
Stop chasing paperwork and wrestling with disconnected systems! Our solution brings clarity and control to your production process. From standard operating procedures (SOPs) and equipment maintenance to quality control and inventory tracking, we simplify every step.
Related Articles
Kaizen Checklist Template: Your Path to Continuous Improvement

Machine Safety Audit Checklist: Your Guide to a Safer Manufacturing Workplace
Mastering Supplier Performance: Your Free Supplier Scorecard Checklist Template
Your Ultimate RCM Checklist Template: A Step-by-Step Guide

LPA Checklist Template: Your Guide to Layered Process Audits

Your Ultimate Lockout/Tagout (LOTO) Checklist Template

Your Ultimate FSC 22000 Checklist Template
Quality Control Inspection Checklist Template: Prevent Defects & Improve Manufacturing Processes
We can do it Together
Need help with
Checklists?
Have a question? We're here to help. Please submit your inquiry, and we'll respond promptly.